Chapter 8: Q4P (page 318)
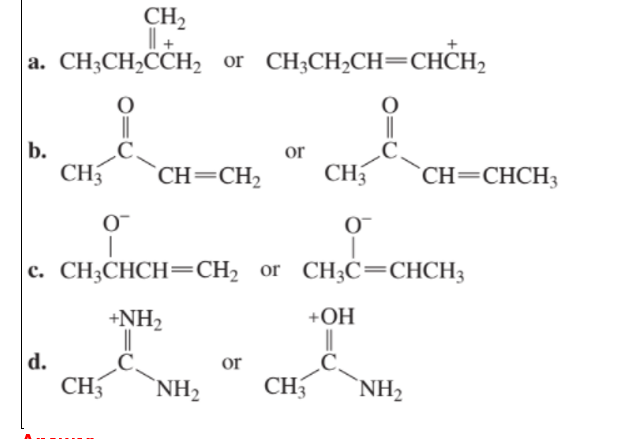
Which species in each pair is more stable?
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q4P (page 318)
Which species in each pair is more stable?


All the tools & learning materials you need for study success - in one app.
Get started for free
Following the instructions for drawing the energy levels of the molecular orbitals for the compounds shown in Figure 8.17, draw the energy levels of the molecular orbitals for the cycloheptatrienyl cation, thecycloheptatrienyl anion, and the cyclopropenyl cation. For each compound, show the distribution of the π electrons. Which of the compounds are aromatic?
Rank the indicated hydrogen in the following compounds from most acidic to least acidic:
Explain why the following compounds are not optically active:
a. the product obtained from the reaction of 1,3-butadiene with cis-1,2-dichloroethene
b. the product obtained from the reaction of 1,3-butadiene with trans-1,2-dichloroethene
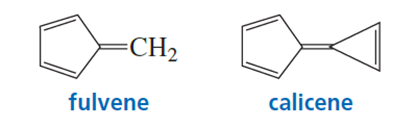
a. In what direction is the dipole moment in fulvene? Explain.
b. In what direction is the dipole moment in calicene? Explain.
The acid dissociation constant (Ka) for loss of a proton from cyclohexanol is 1 ×10-16.
a. Draw a reaction coordinate diagram for loss of a proton from cyclohexanol.
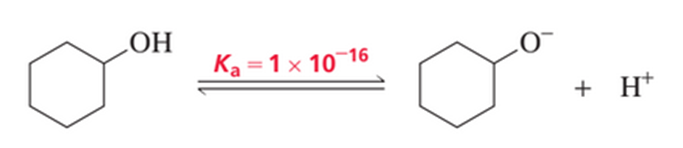
b. Draw the resonance contributors for phenol.
c. Draw the resonance contributors for the phenolate ion.
d. On the same plot with the energy diagram for loss of a proton from cyclohexanol, draw an energy diagram for loss of a proton from phenol.
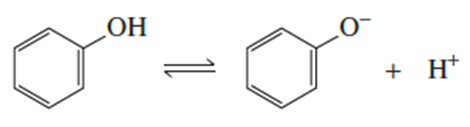
e. Which has a greater Ka: cyclohexanol or phenol?
f. Which is a stronger acid: cyclohexanol or phenol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.