Chapter 8: Q49P (page 364)
a.What is the value of nin Hückel’s rule when a compound has nine pairs of π electrons?
b. Is such a compound aromatic?
Short Answer
(a)The value of n will be 4.
(b) The compound is aromatic.
Learning Materials
Features
Discover
Chapter 8: Q49P (page 364)
a.What is the value of nin Hückel’s rule when a compound has nine pairs of π electrons?
b. Is such a compound aromatic?
(a)The value of n will be 4.
(b) The compound is aromatic.
All the tools & learning materials you need for study success - in one app.
Get started for free
Which has the greater delocalization energy?
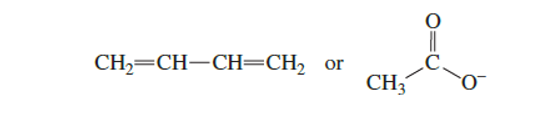
a. The following compounds have the same molecular formula as benzene. How many monosubstituted products would each have?
1.���≡C��≡C���2CH3 2. CH2≡C���≡C���≡C��2
b. How many disubstituted products would each of the preceding compounds have? (Do not include stereoisomers.)
c. How many disubstituted products would each of the compounds have if stereoisomers are included?
Which of the following compounds could be protonated without destroying its aromaticity?
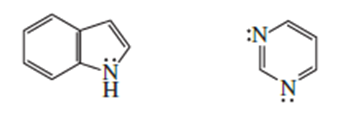
Which of the following is the strongest acid?
a. How could each of the following compounds be prepared from a hydrocarbon in a single step?
b. What other organic compound would be obtained from each synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.