Chapter 8: Q90P (page 379)
a. How could each of the following compounds be prepared from a hydrocarbon in a single step?
b. What other organic compound would be obtained from each synthesis?
Learning Materials
Features
Discover
Chapter 8: Q90P (page 379)
a. How could each of the following compounds be prepared from a hydrocarbon in a single step?
b. What other organic compound would be obtained from each synthesis?
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw resonance contributors for the following species, showing all the lone pairs:
1.CH2N2 2. N2O 3. NO2-
b. For each species, indicate the most stable resonance contributor.
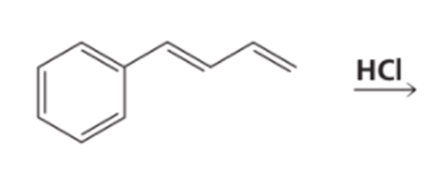
Identify the kinetic and thermodynamic products of the following reaction:
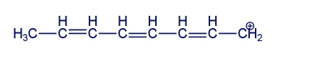
Draw the resonance contributors for the following carbocation
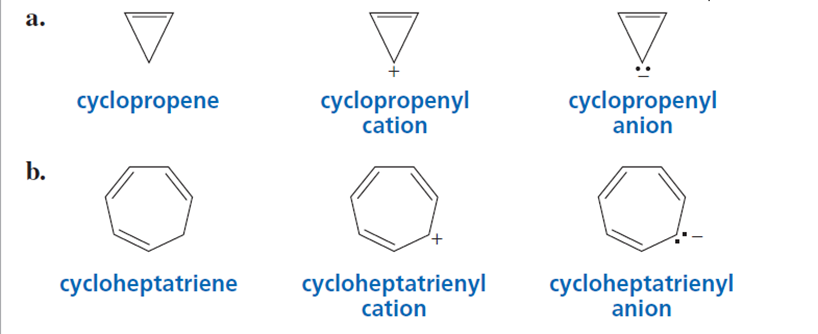
Which compound in each set is aromatic? Explain your choice.
The heat of hydrogenation of 2,3-pentadiene, a cumulated diene, is 70.5 kcal/mol. What are the relative stabilities of cumulated, conjugated, and isolated dienes?
What do you think about this solution?
We value your feedback to improve our textbook solutions.