Chapter 8: Q50P (page 365)
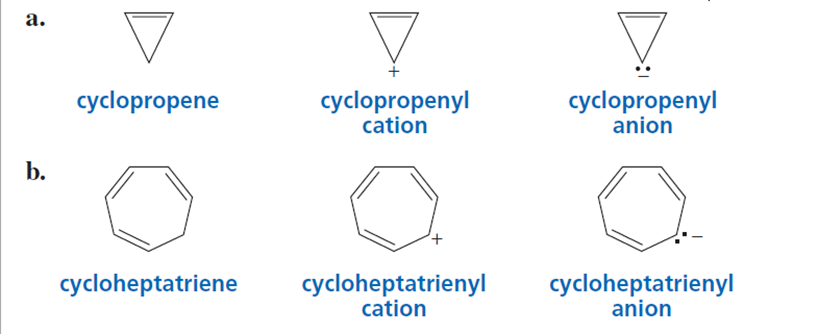
Which compound in each set is aromatic? Explain your choice.
Short Answer
(a) From the 1st set, Cyclopropenyl cation is aromatic.
(b) From the 2nd set, Cycloheptatrienyl cation is aromatic.
Learning Materials
Features
Discover
Chapter 8: Q50P (page 365)
Which compound in each set is aromatic? Explain your choice.

(a) From the 1st set, Cyclopropenyl cation is aromatic.
(b) From the 2nd set, Cycloheptatrienyl cation is aromatic.
All the tools & learning materials you need for study success - in one app.
Get started for free
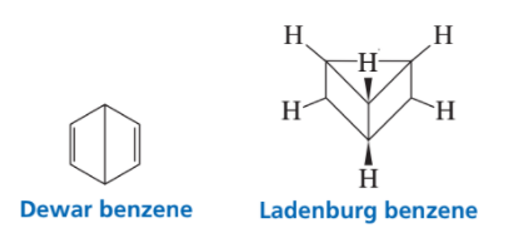
Between 1865 and 1890, other possible structures were proposed for benzene such as those shown here. Considering what nineteenth-century chemists knew about benzene, which is a better proposal for benzene’s structure, Dewar benzene or Ladenburg benzene? Why?
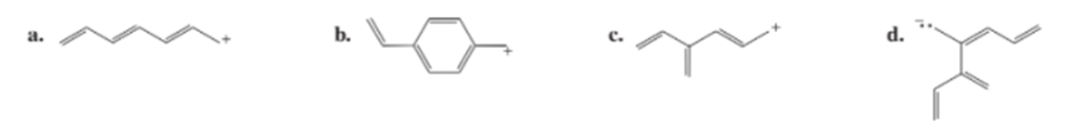
Draw resonance contributors for the following ions:
What orbital do the lone-pair electrons occupy in each of the following compounds?
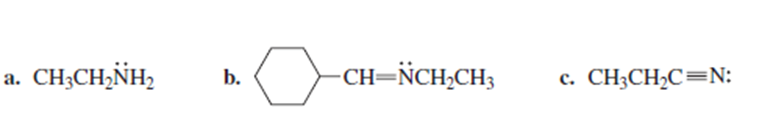
a. When HBr adds to a conjugated diene, what is the rate-determining step?
b. When HBr adds to a conjugated diene, what is the product-determining step?
A student obtained two products from the reaction of 1,3-cyclohexadiene with Br2 (disregarding stereoisomers). His lab partner was surprised when he obtained only one product from the reaction of 1,3-cyclohexadiene with HBr (disregarding stereoisomers). Account for these results.
What do you think about this solution?
We value your feedback to improve our textbook solutions.