Chapter 8: 8-1 TP (page 381)
Draw the resonance contributors for the following carbocation
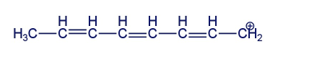
Short Answer
Learning Materials
Features
Discover
Chapter 8: 8-1 TP (page 381)
Draw the resonance contributors for the following carbocation


All the tools & learning materials you need for study success - in one app.
Get started for free
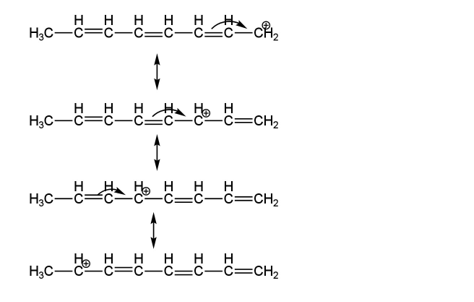
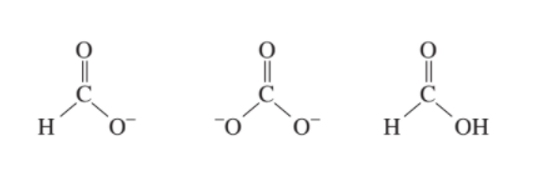
Draw resonance contributors for each of the following species and rank them in order of decreasing contribution to the hybrid. Then draw the resonance hybrid.
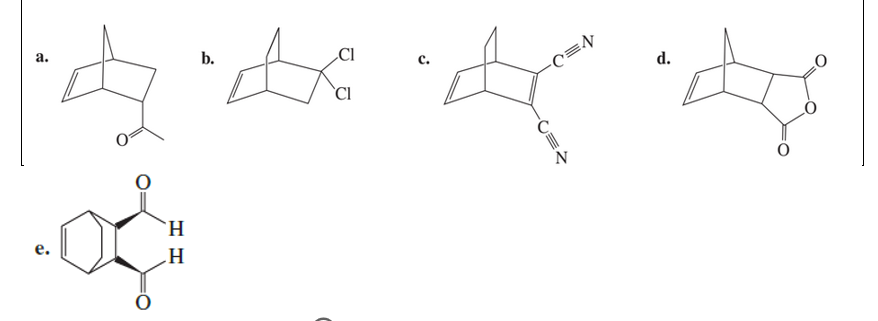
Which diene and which dienophile could be used to prepare each of the following?
List the following in order of decreasing delocalization energy:
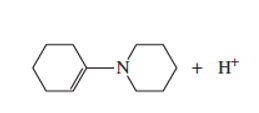
Which atom in the following compound is most likely to be protonated?
The A ring ( Section 3.16 ) of cortisone, a steroid, is formed by a Diels–Alder reaction using the two reactants shown here. What is the product of this reaction?
The C ring of estrone (a steroid) is formed by a Diels–Alder reaction using the tworeactants shown here. What is the product of this reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.