Chapter 8: Q26P (page 318)
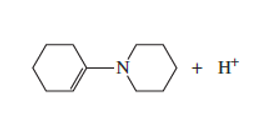
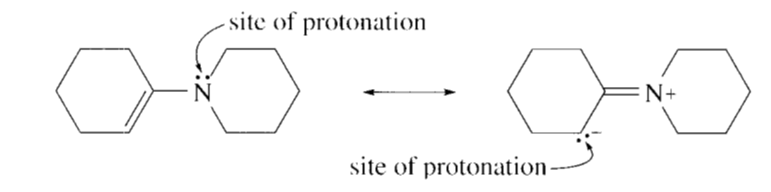
Which atom in the following compound is most likely to be protonated?
Short Answer
Two sites can be protonated i.e. lone pair on nitrogen and lone pair on carbon.
Learning Materials
Features
Discover
Chapter 8: Q26P (page 318)
Which atom in the following compound is most likely to be protonated?

Two sites can be protonated i.e. lone pair on nitrogen and lone pair on carbon.

All the tools & learning materials you need for study success - in one app.
Get started for free
Which ion in each of the following pairs is more stable?

Refer to the electrostatic potential maps on p. 368 to answer the following questions:
a. Why is the bottom part of the electrostatic potential map of pyrrole blue?
b. Why is the bottom part of the electrostatic potential map of pyridine red?
c. Why is the center of the electrostatic potential map of benzene more red than the center of the electrostatic
potential map of pyridine?
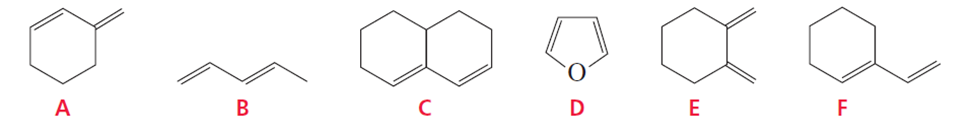
Which of the following conjugated dienes will not react with a dienophile in a Diels–Alder reaction?
Explain why the following compounds are not optically active:
a. the product obtained from the reaction of 1,3-butadiene with cis-1,2-dichloroethene
b. the product obtained from the reaction of 1,3-butadiene with trans-1,2-dichloroethene
Following the instructions for drawing the energy levels of the molecular orbitals for the compounds shown in Figure 8.17, draw the energy levels of the molecular orbitals for the cycloheptatrienyl cation, thecycloheptatrienyl anion, and the cyclopropenyl cation. For each compound, show the distribution of the π electrons. Which of the compounds are aromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.