Chapter 8: Q27P (page 318)
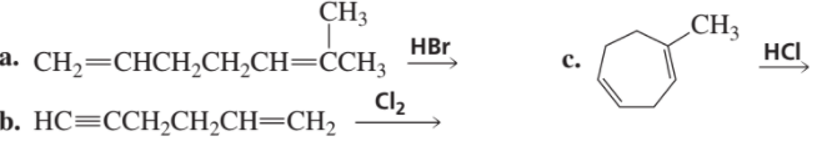
What is the major product of each of the following reactions, assuming that one equivalent of each reagent is used in each reaction?
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q27P (page 318)
What is the major product of each of the following reactions, assuming that one equivalent of each reagent is used in each reaction?


All the tools & learning materials you need for study success - in one app.
Get started for free
A student wanted to know whether the greater proximity of the nucleophile to the C-2 carbon in the transition state is what causes the 1,2 addition product to be formed faster when 1,3-butadiene reacts with HCl. Therefore, she decided to investigate the reaction of 2-methyl-1,3 cyclohexadiene with HCl. Her friend told her that she should use 1-methyl-1,3 cyclohexadiene instead. Should she follow her friend’s advice?
a) Propose a mechanism for the following reaction:
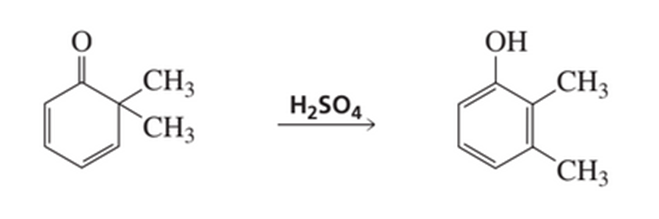
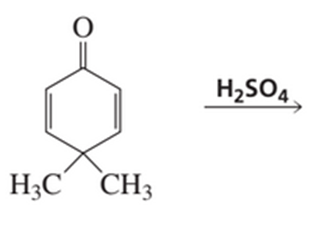
b) What is the product of the following reaction?
Which dienophile in each pair is more reactive in a Diels–Alder reaction?
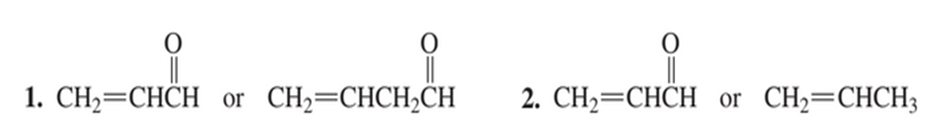
Which diene is more reactive in a Diels–Alder reaction?
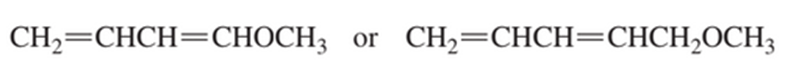
Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene:
a. How many MOs does the compound have?
b. Which are the bonding MOs, and which are the antibonding MOs?
c. Which MOs are symmetric, and which are antisymmetric?
d. Which MO is the HOMO and which is the LUMO in the ground state?
e. Which MO is the HOMO and which is the LUMO in the excited state?
f. What is the relationship between HOMO and LUMO and symmetric and antisymmetric orbitals?
g. How many nodes does the highest-energy MO of 1,3,5,7-octatetraene have between the nuclei?
Which compound would you expect to have the greater heat of hydrogenation: 1,2-pentadiene or 1,4-pentadiene?
What do you think about this solution?
We value your feedback to improve our textbook solutions.