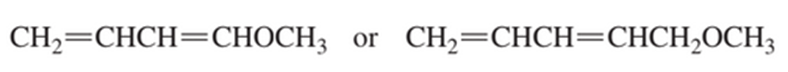Chapter 8: Q86P (page 379)
Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene:
a. How many MOs does the compound have?
b. Which are the bonding MOs, and which are the antibonding MOs?
c. Which MOs are symmetric, and which are antisymmetric?
d. Which MO is the HOMO and which is the LUMO in the ground state?
e. Which MO is the HOMO and which is the LUMO in the excited state?
f. What is the relationship between HOMO and LUMO and symmetric and antisymmetric orbitals?
g. How many nodes does the highest-energy MO of 1,3,5,7-octatetraene have between the nuclei?
Short Answer
a. 8
��.𝛹1, 𝛹2, 𝛹3, 𝛹4 are bonding MOs and 𝛹5 ,𝛹6 ,𝛹7, 𝛹 8 are antibonding MOs.
c. 𝛹5 ,𝛹6 ,𝛹7 ,𝛹 8are symmetric and 𝛹2, 𝛹4 ,𝛹6, 𝛹8 are antisymmetric.
d. 𝛹4 is HOMO and 𝛹 5is LUMO at the ground state.
e. 𝛹 5is HOMO and 𝛹6 is LUMO in the excited state.
f. If the HOMO is symmetric then LUMO will be asymmetric and vice versa.
g. 7 nodes.