Chapter 8: Q87P (page 379)
How could you synthesize the following compound from starting materials containing no more than six carbons? (Hint: A 1,6-diketone can be synthesized by oxidative cleavage of a 1,2-disubstituted cyclohexene.)
Learning Materials
Features
Discover
Chapter 8: Q87P (page 379)
How could you synthesize the following compound from starting materials containing no more than six carbons? (Hint: A 1,6-diketone can be synthesized by oxidative cleavage of a 1,2-disubstituted cyclohexene.)
All the tools & learning materials you need for study success - in one app.
Get started for free
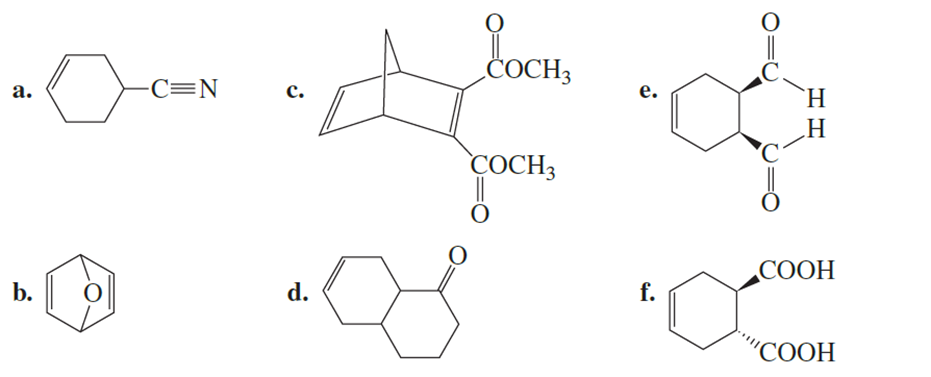
What diene and what dienophile should be used to synthesize the following?
What two products are formed from each of the following reactions?
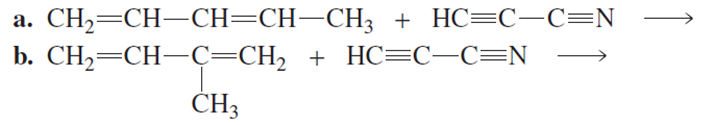
The acid dissociation constant (Ka) for loss of a proton from cyclohexanol is 1 ×10-16.
a. Draw a reaction coordinate diagram for loss of a proton from cyclohexanol.
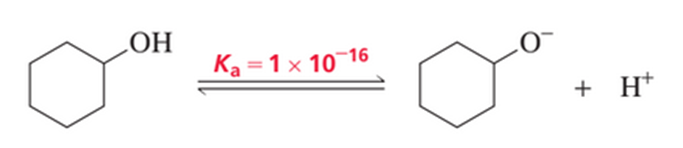
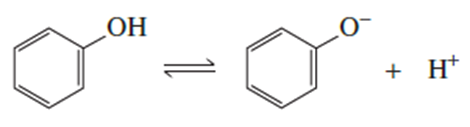
b. Draw the resonance contributors for phenol.
c. Draw the resonance contributors for the phenolate ion.
d. On the same plot with the energy diagram for loss of a proton from cyclohexanol, draw an energy diagram for loss of a proton from phenol.
e. Which has a greater Ka: cyclohexanol or phenol?
f. Which is a stronger acid: cyclohexanol or phenol?
The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated and stored. Why is this carbocation so stable?
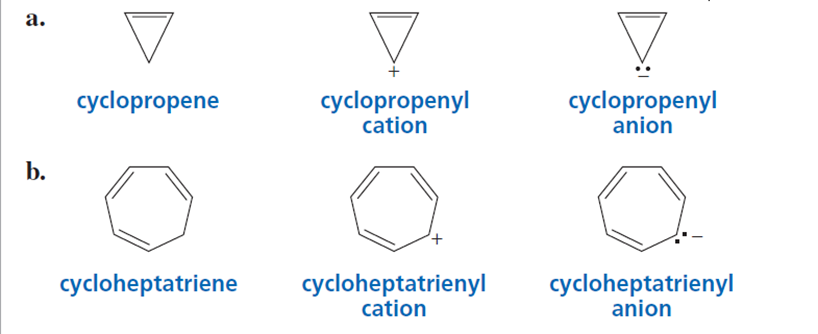
Which compound in each set is aromatic? Explain your choice.
What do you think about this solution?
We value your feedback to improve our textbook solutions.