Chapter 8: Q47P (page 362)
What diene and what dienophile should be used to synthesize the following?
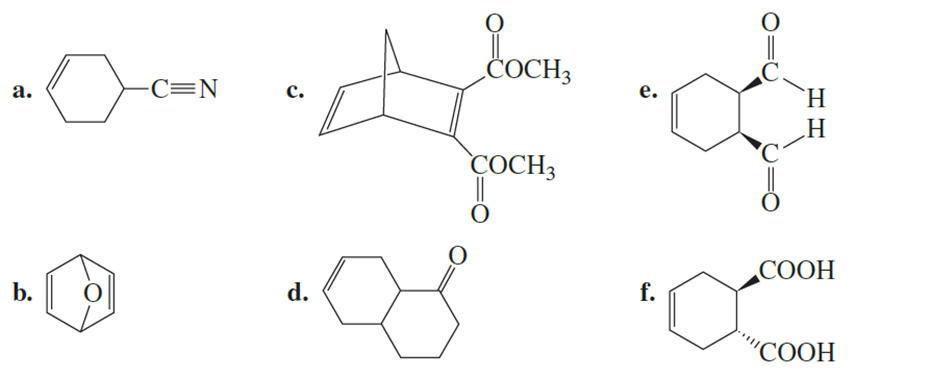
Short Answer
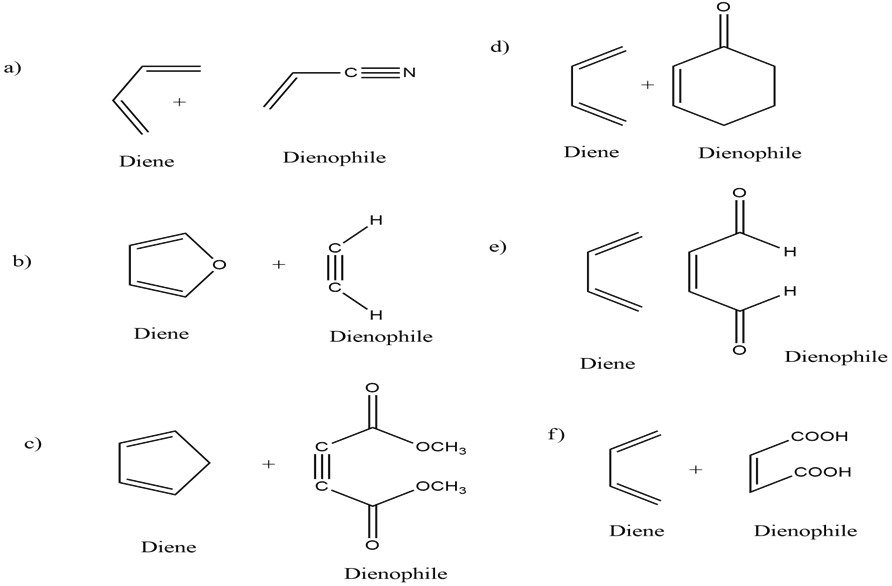
The diene and dienophile for all of the reactions are,
Learning Materials
Features
Discover
Chapter 8: Q47P (page 362)
What diene and what dienophile should be used to synthesize the following?

The diene and dienophile for all of the reactions are,

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Why does deuterium add to C-1 rather than to C-4 in the preceding reaction?
b. Why was DCl rather than HCl used in the reaction?
a. How many linear dienes have molecular formula C6H10? (Disregard cis-trans isomers.)
b.How many of the linear dienes in part a are conjugated dienes?
c.How many are isolated dienes?
d. How many are cumulated dienes?
Refer to the electrostatic potential maps on p. 368 to answer the following questions:
a. Why is the bottom part of the electrostatic potential map of pyrrole blue?
b. Why is the bottom part of the electrostatic potential map of pyridine red?
c. Why is the center of the electrostatic potential map of benzene more red than the center of the electrostatic
potential map of pyridine?
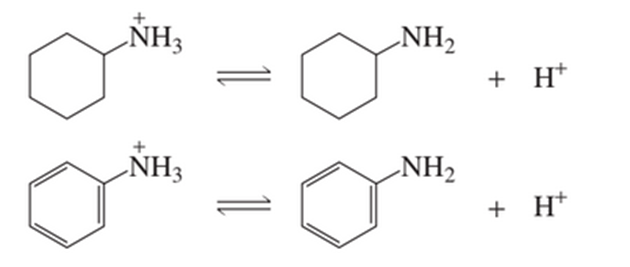
Protonated cyclohexylamine has aKa=1×10−11. Using the same sequence of steps as in Problem 94 , determine which is a stronger base, cyclohexylamine or aniline.
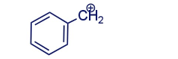
Draw the resonance contributors for the following carbocation
What do you think about this solution?
We value your feedback to improve our textbook solutions.