Chapter 8: Q33 P (page 353)
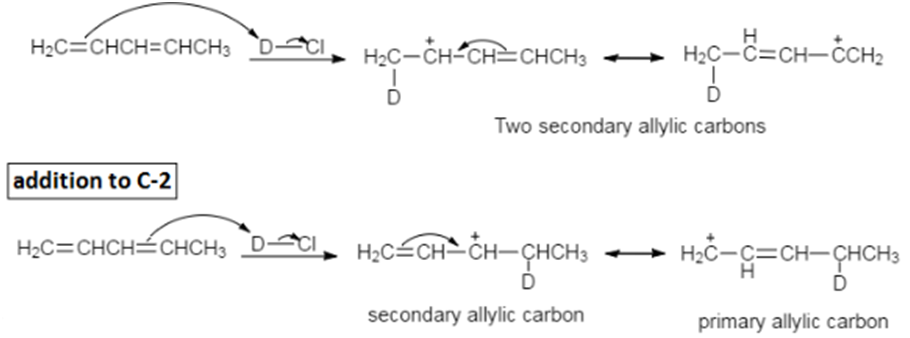
a. Why does deuterium add to C-1 rather than to C-4 in the preceding reaction?
b. Why was DCl rather than HCl used in the reaction?
Short Answer
a) Addition at C-1 forms the most stable carbocation because the positive charge is shared by two secondary allylic carbons. If the deuterium had added to C-4 the positive charge would have been shared by a secondary and a primary allylic carbon.
b) DCl was used to cause the 1,2- and 1,4- products to be different. If HCl had been used, the 1,2- and 1,4- products would have been the same.