Chapter 8: Q46 P (page 361)
Explain why the following compounds are not optically active:
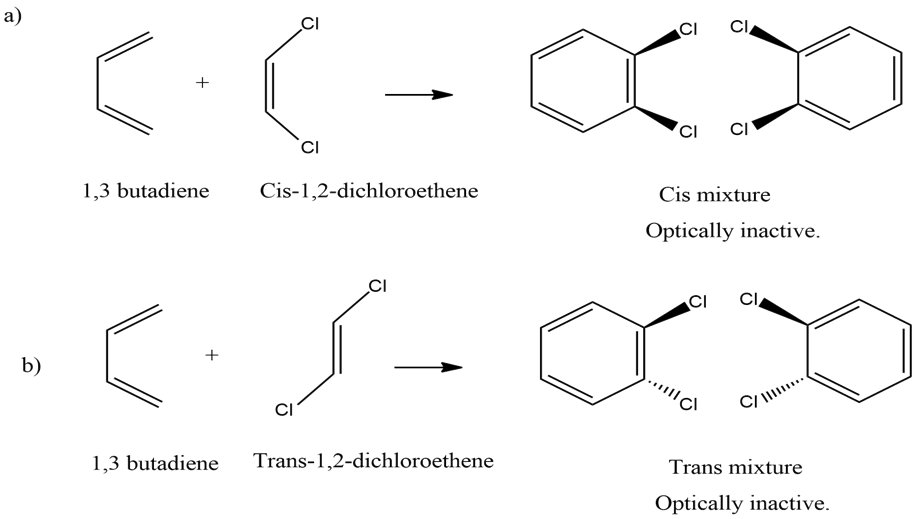
a. the product obtained from the reaction of 1,3-butadiene with cis-1,2-dichloroethene
b. the product obtained from the reaction of 1,3-butadiene with trans-1,2-dichloroethene
Short Answer
Diels -Alder reaction is a syn addition reaction. So, each of the following syn addition reactions forms a product with two new asymmetric centers with forms a pair of enantiomers. Now as enantiomers are a mixture of two opposite optically active compounds, the overall optical activity of the mixture get nullified.