Chapter 8: Q48 P (page 364)
Which of the following are aromatic?
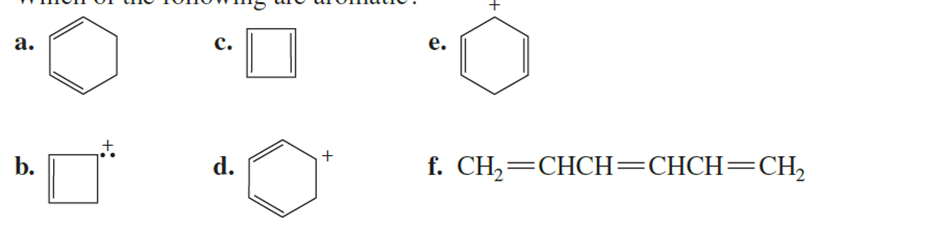
Short Answer
Among all the 6 compounds, compound b and f are aromatic compounds according toHückel’s rule [(4n+ 2) π electrons].
Learning Materials
Features
Discover
Chapter 8: Q48 P (page 364)
Which of the following are aromatic?
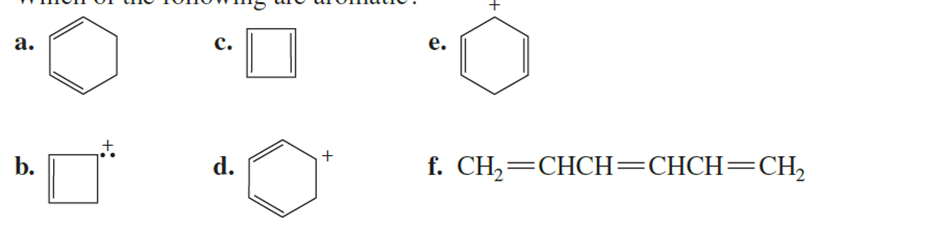
Among all the 6 compounds, compound b and f are aromatic compounds according toHückel’s rule [(4n+ 2) π electrons].
All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound is the strongest base?
Cyclopentadiene can react with itself in a Diels–Alder reaction. Draw the endo and exo products.
The heat of hydrogenation of 2,3-pentadiene, a cumulated diene, is 70.5 kcal/mol. What are the relative stabilities of cumulated, conjugated, and isolated dienes?
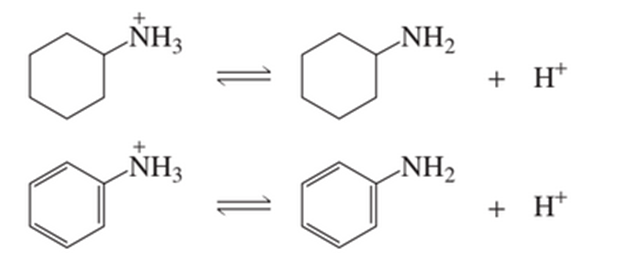
Protonated cyclohexylamine has aKa=1×10−11. Using the same sequence of steps as in Problem 94 , determine which is a stronger base, cyclohexylamine or aniline.
How could you synthesize the following compound from starting materials containing no more than six carbons? (Hint: A 1,6-diketone can be synthesized by oxidative cleavage of a 1,2-disubstituted cyclohexene.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.