Chapter 8: Q74P (page 377)
Which compound is the strongest base?
Short Answer
This is the strongest base.
Learning Materials
Features
Discover
Chapter 8: Q74P (page 377)
Which compound is the strongest base?
This is the strongest base.
All the tools & learning materials you need for study success - in one app.
Get started for free
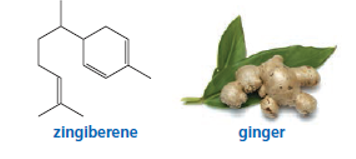
Which of the double bonds in zingiberene, the compound responsible for the aroma of ginger, is most reactive in an electrophilic addition reaction with HBr?
Draw the resonance contributors of the cyclooctatrienyl dianion.
a. Which of the resonance contributors is the least stable?
b. Which of the resonance contributors makes the smallest contribution to the hybrid?
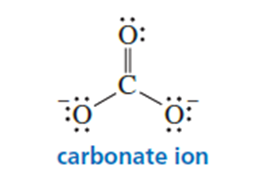
a. Predict the relative bond lengths of the three carbon-oxygen bonds in carbonate ion.
b. What is the charge on each oxygen?
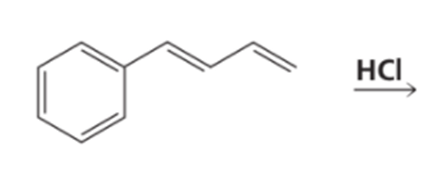
Identify the kinetic and thermodynamic products of the following reaction:
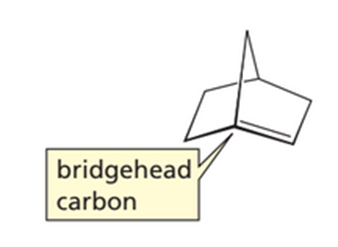
In 1935, J. Bredt, a German chemist, proposed that a bicycloalkene could not have a double bond at a bridgehead carbon unless one of the rings contains at least eight carbons. This is known as Bredt’s rule. Explain why there cannot be a double bond at this position.
What do you think about this solution?
We value your feedback to improve our textbook solutions.