Chapter 8: Q75P (page 377)
Which loses a proton more readily: a methyl group bonded to cyclohexane or a methyl group bonded to benzene?
Short Answer
The methyl group on benzene can lose a proton easier than the methyl group on cyclohexane.
Learning Materials
Features
Discover
Chapter 8: Q75P (page 377)
Which loses a proton more readily: a methyl group bonded to cyclohexane or a methyl group bonded to benzene?
The methyl group on benzene can lose a proton easier than the methyl group on cyclohexane.
All the tools & learning materials you need for study success - in one app.
Get started for free
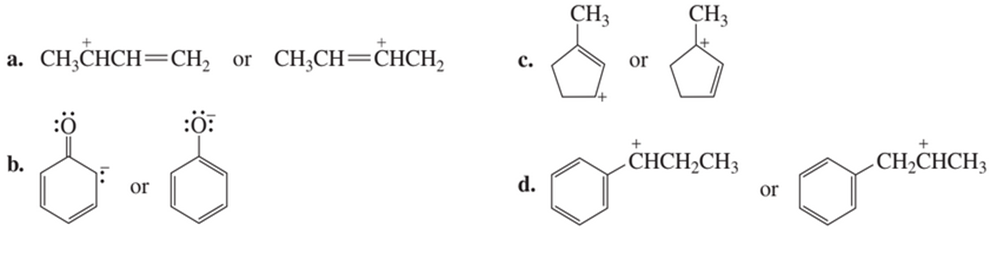
Which resonance contributor in each pair makes the greater contribution to the resonance hybrid?
The heat of hydrogenation of 2,3-pentadiene, a cumulated diene, is 70.5 kcal/mol. What are the relative stabilities of cumulated, conjugated, and isolated dienes?
Draw the resonance contributors for the following carbocation
a) What are the products of the following reaction?
b) How many stereoisomers of each product could be obtained?
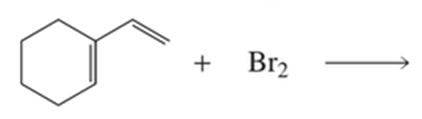
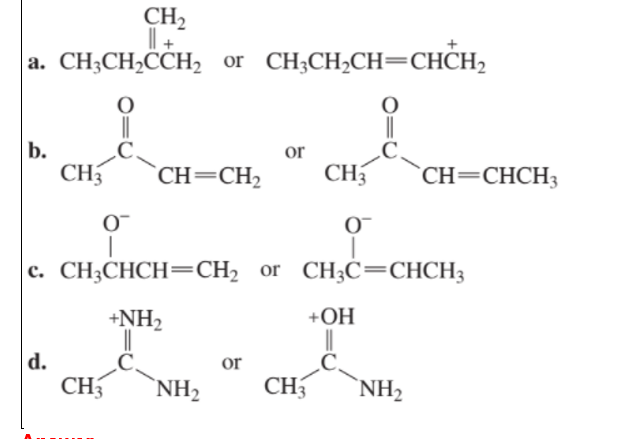
Which species in each pair is more stable?
What do you think about this solution?
We value your feedback to improve our textbook solutions.