Chapter 8: Q42 P (page 360)
What two products are formed from each of the following reactions?
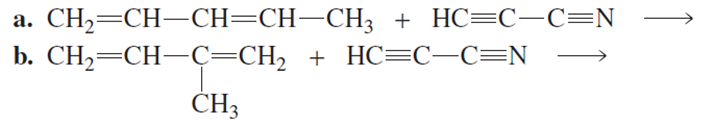
Short Answer
The following products are given below following the Diels-Alder reaction.
Learning Materials
Features
Discover
Chapter 8: Q42 P (page 360)
What two products are formed from each of the following reactions?
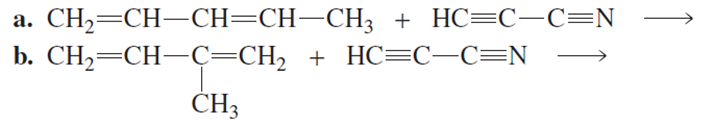
The following products are given below following the Diels-Alder reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Write a general rule that can be used to predict the major product of a Diels–Alder reaction between an alkene with an electron-withdrawing substituent and a diene with a substituent that can donate electrons by resonance depending on the location of the substituent on the diene.
What is the major product obtained from the addition of hbr to the following compound.
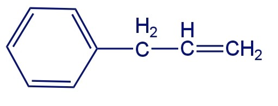
Draw the major products obtained from the reaction of one equivalent of HCl with the following compounds. For each reaction, indicate the kinetic and thermodynamic products.
a. 2,3-dimethyl-1,3-pentadiene
b. 2,4-dimethyl-1,3-pentadiene
Which species in each of the pairs in Problem 80 is the stronger base?
The experiment shown next and discussed in Section 8.13 shows that the proximity of the chloride ion to C−2 in the transition state causes the 1,2−addition product to be formed faster than the 1,4−addition product.
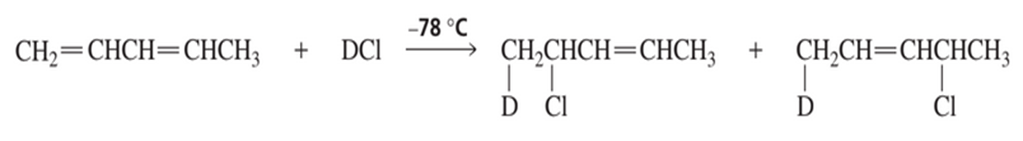
a. Why was it important for the investigators to know that the preceding reaction was being carried out under kinetic control?
b. How could the investigators know that the reaction was being carried out under kinetic control?
What do you think about this solution?
We value your feedback to improve our textbook solutions.