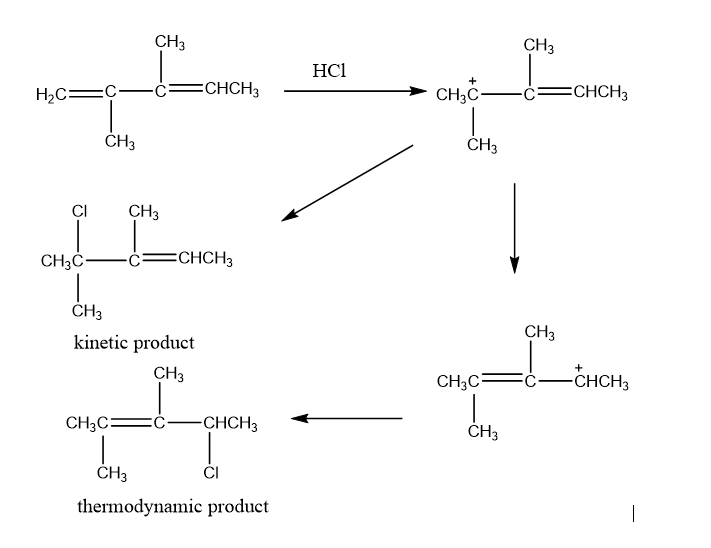
Chapter 8: 8-93P (page 379)
Draw the major products obtained from the reaction of one equivalent of HCl with the following compounds. For each reaction, indicate the kinetic and thermodynamic products.
a. 2,3-dimethyl-1,3-pentadiene
b. 2,4-dimethyl-1,3-pentadiene
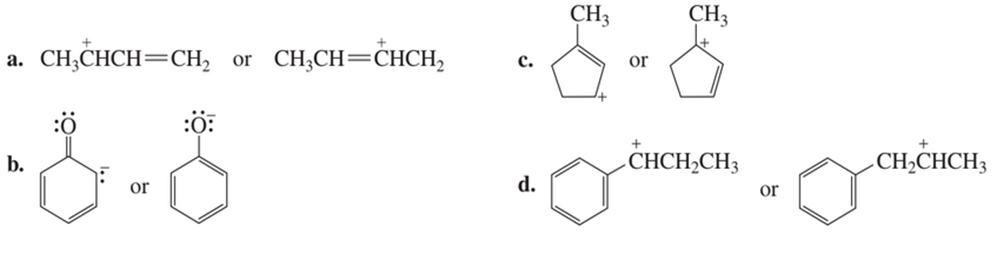
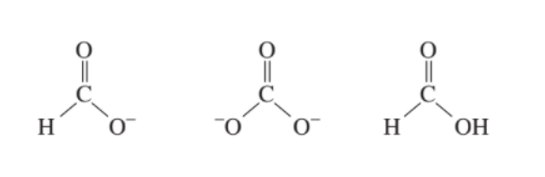
Short Answer