Chapter 8: Q85P (page 378)
Rank the indicated hydrogen in the following compounds from most acidic to least acidic:
Short Answer
The order of acidity is - a> b> c
Learning Materials
Features
Discover
Chapter 8: Q85P (page 378)
Rank the indicated hydrogen in the following compounds from most acidic to least acidic:
The order of acidity is - a> b> c
All the tools & learning materials you need for study success - in one app.
Get started for free
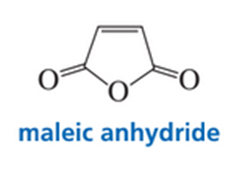
While attempting to recrystallize maleic anhydride, a student dissolved it in freshly distilled cyclopentadiene rather than in freshly distilled cyclopentane. Was his recrystallization successful?
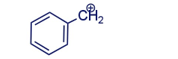
Draw the resonance contributors for the following carbocation
Explain why the pka of p-nitrophenol is 7.14, whereas the pka of m-nitrophenol is 8.39.
What is the major product of each of the following reactions? Assume that there is an equivalent amount of each reagent.

Following the instructions for drawing the energy levels of the molecular orbitals for the compounds shown in Figure 8.17, draw the energy levels of the molecular orbitals for the cycloheptatrienyl cation, thecycloheptatrienyl anion, and the cyclopropenyl cation. For each compound, show the distribution of the π electrons. Which of the compounds are aromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.