Chapter 8: Q 69P (page 376)
Which ion in each of the following pairs is more stable?

Short Answer
Here we have to find out more stable compound.
a)
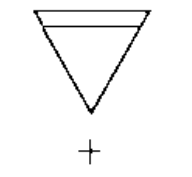
b)
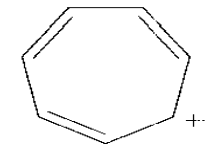
c)
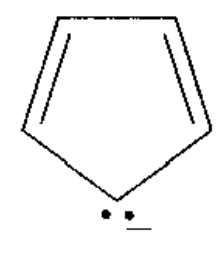
d)
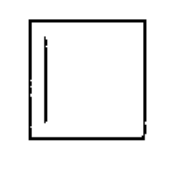
Learning Materials
Features
Discover
Chapter 8: Q 69P (page 376)
Which ion in each of the following pairs is more stable?

Here we have to find out more stable compound.
a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Which loses a proton more readily: a methyl group bonded to cyclohexane or a methyl group bonded to benzene?
The following equilibrium is driven to the right if the reaction is carried out in the presence of maleic anhydride (see Problem 106). What is the function of maleic anhydride?
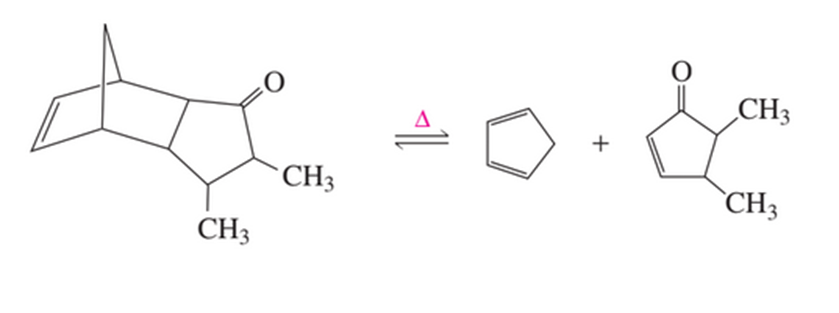
What are the products of the following reactions, assuming that one equivalent of each reagent is used in each reaction?
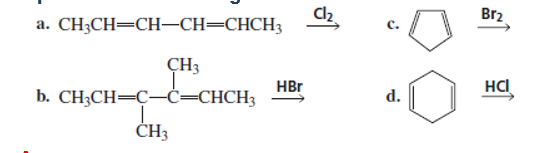
A student wanted to know whether the greater proximity of the nucleophile to the C-2 carbon in the transition state is what causes the 1,2 addition product to be formed faster when 1,3-butadiene reacts with HCl. Therefore, she decided to investigate the reaction of 2-methyl-1,3 cyclohexadiene with HCl. Her friend told her that she should use 1-methyl-1,3 cyclohexadiene instead. Should she follow her friend’s advice?
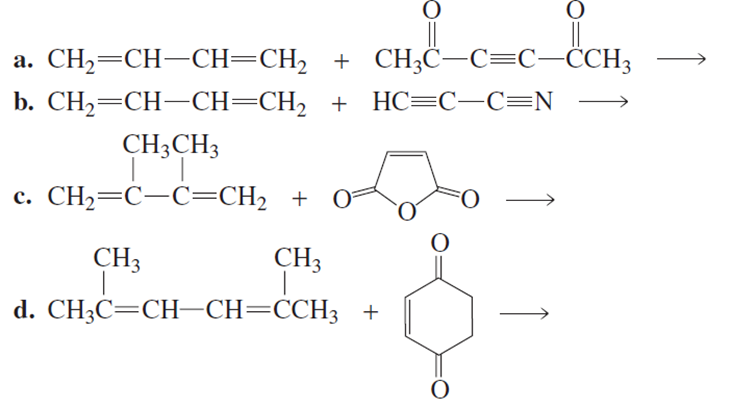
What are the products of the following reactions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.