Chapter 8: Q54P (page 367)
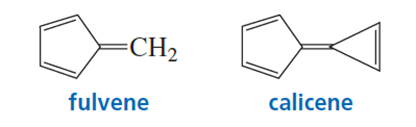
a. In what direction is the dipole moment in fulvene? Explain.
b. In what direction is the dipole moment in calicene? Explain.
Short Answer
The direction of dipole moment are given below for those compounds,

Learning Materials
Features
Discover
Chapter 8: Q54P (page 367)
a. In what direction is the dipole moment in fulvene? Explain.
b. In what direction is the dipole moment in calicene? Explain.

The direction of dipole moment are given below for those compounds,

All the tools & learning materials you need for study success - in one app.
Get started for free
Are the following pairs of structures resonance contributors or different compounds?

Which species in each pair is more stable?
Write a general rule that can be used to predict the major product of a Diels–Alder reaction between an alkene with an electron-withdrawing substituent and a diene with a substituent that can donate electrons by resonance depending on the location of the substituent on the diene.
What products would be obtained from the reaction of 1,3,5-hexatriene with one equivalent of HBr? Disregard stereoisomers.
a.What is the value of nin Hückel’s rule when a compound has nine pairs of π electrons?
b. Is such a compound aromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.