Chapter 8: Q 66P (page 376)
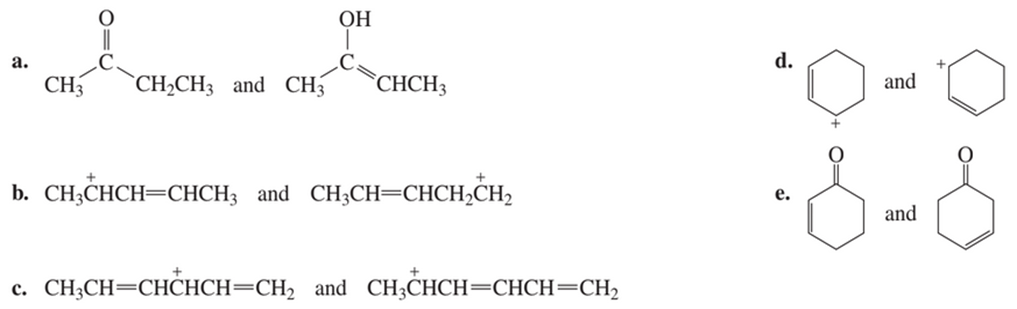
Are the following pairs of structures resonance contributors or different compounds?
Short Answer
a) Different compounds
b) Different compounds
c) Resonance contributors
d) Resonance contributors
e) Different compounds
Learning Materials
Features
Discover
Chapter 8: Q 66P (page 376)
Are the following pairs of structures resonance contributors or different compounds?

a) Different compounds
b) Different compounds
c) Resonance contributors
d) Resonance contributors
e) Different compounds
All the tools & learning materials you need for study success - in one app.
Get started for free
What stereoisomers do the first two reactions on the top of p. 348 form? (Hint: Review Section 6.13.)
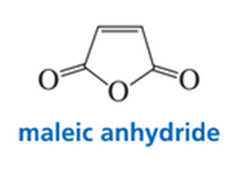
While attempting to recrystallize maleic anhydride, a student dissolved it in freshly distilled cyclopentadiene rather than in freshly distilled cyclopentane. Was his recrystallization successful?
What orbitals contain the electrons represented as lone pairs in the structures of quinoline, indole, imidazole, purine, and pyrimidine?
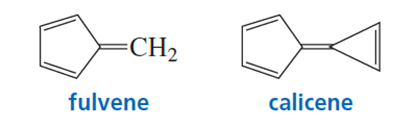
a. In what direction is the dipole moment in fulvene? Explain.
b. In what direction is the dipole moment in calicene? Explain.
Which compound in each set is aromatic? Explain your choice.
What do you think about this solution?
We value your feedback to improve our textbook solutions.