Chapter 8: Q57P (page 369)
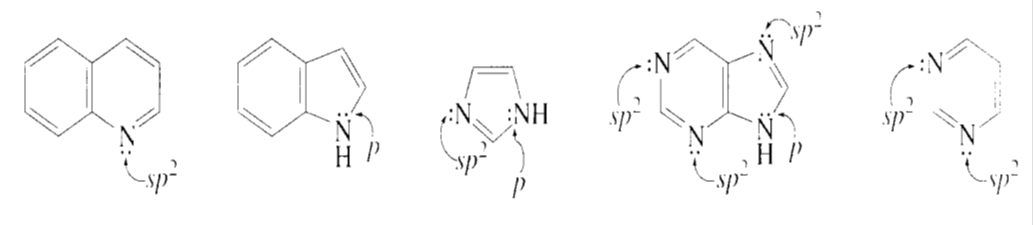
What orbitals contain the electrons represented as lone pairs in the structures of quinoline, indole, imidazole, purine, and pyrimidine?
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q57P (page 369)
What orbitals contain the electrons represented as lone pairs in the structures of quinoline, indole, imidazole, purine, and pyrimidine?

All the tools & learning materials you need for study success - in one app.
Get started for free
On a single graph, draw the reaction coordinate diagram for the addition of one equivalent of HBr to 2 methyl-1,3-pentadiene and for the addition of one equivalent of HBr to 2-methyl-1,4-pentadiene. Which reaction is faster?
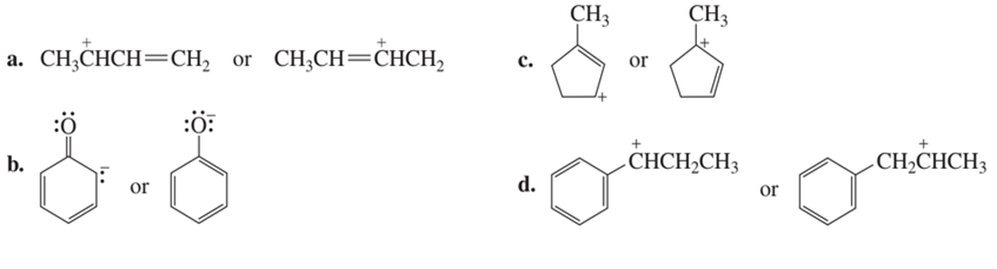
Which resonance contributor in each pair makes the greater contribution to the resonance hybrid?
What orbital do the lone-pair electrons occupy in each of the following compounds?
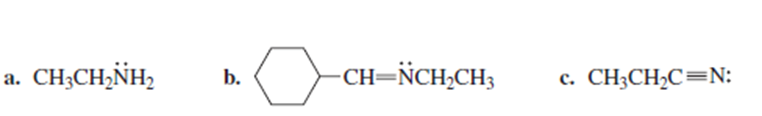
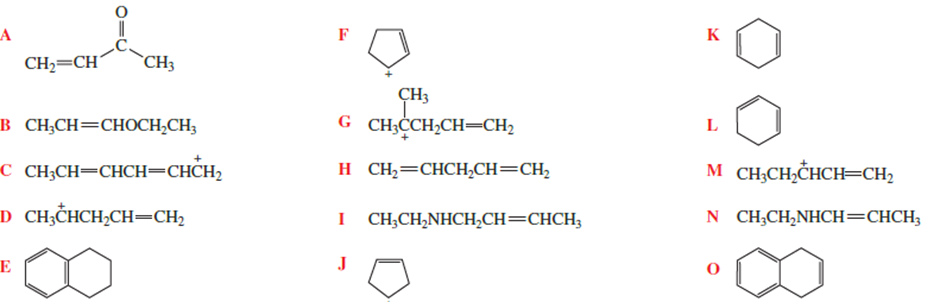
Which of the following has delocalized electrons?
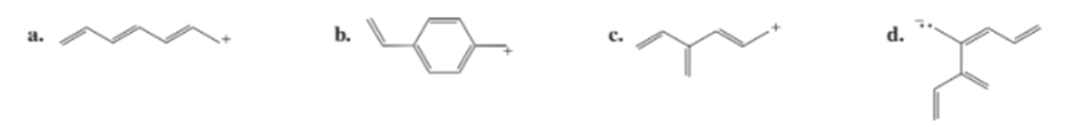
Draw resonance contributors for the following ions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.