Chapter 8: Q53P (page 366)
Which is more soluble in water, 3-bromocyclopropene or bromocyclopropane?
Short Answer
3-bromocyclopropene will be more soluble in water than bromocyclopropane.
Learning Materials
Features
Discover
Chapter 8: Q53P (page 366)
Which is more soluble in water, 3-bromocyclopropene or bromocyclopropane?
3-bromocyclopropene will be more soluble in water than bromocyclopropane.
All the tools & learning materials you need for study success - in one app.
Get started for free
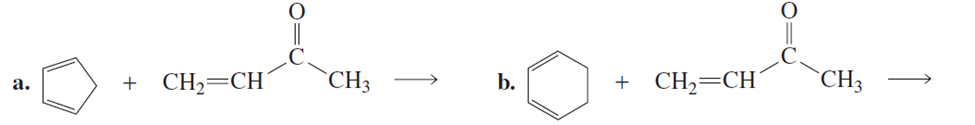
What is the major product of each of the following reactions? Assume that there is an equivalent amount of each reagent.

a. Which oxygen atom has the greater electron density?
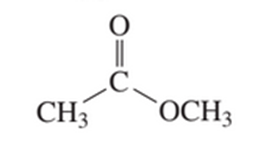
b. Which compound has the greater electron density on its nitrogen atom?
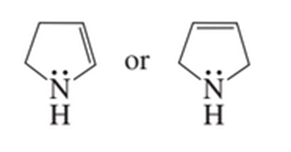
c. Which compound has the greater electron density on its oxygen atom?
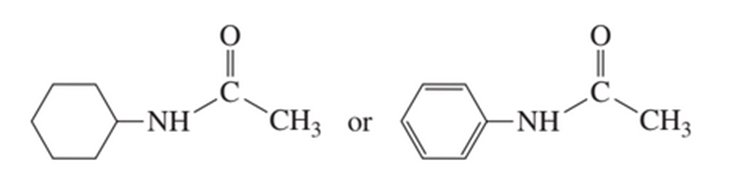
What are the products of the following reactions?
Which carbocation in each pair is more stable?
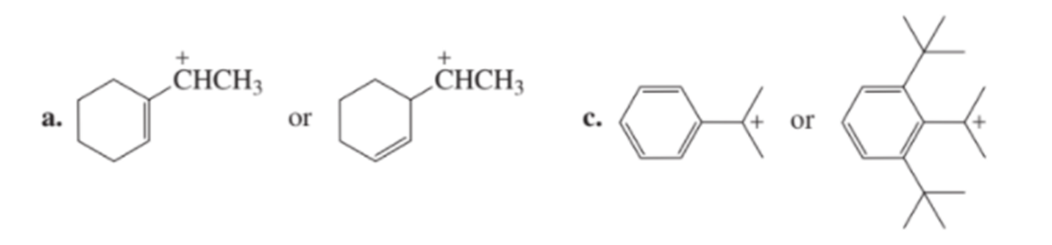
b. CH3OC+H2 or CH3NHC+H2
How would the following substituents affect the rate of a Diels–Alder reaction?
a. an electron-donating substituent in the diene
b. an electron-donating substituent in the dienophile
c. an electron-withdrawing substituent in the diene
What do you think about this solution?
We value your feedback to improve our textbook solutions.