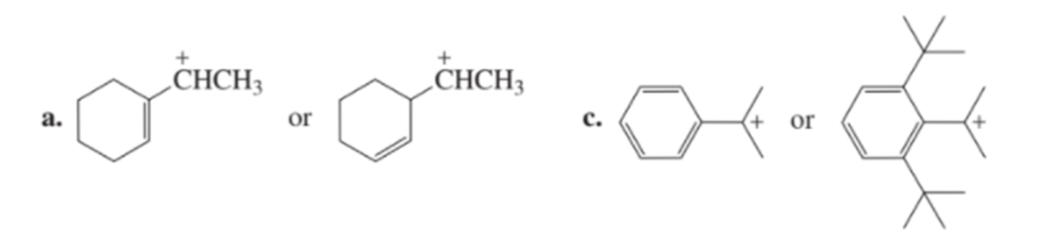
Chapter 8: Q13P (page 334)
Which carbocation in each pair is more stable?
b. CH3OC+H2 or CH3NHC+H2
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q13P (page 334)
Which carbocation in each pair is more stable?

b. CH3OC+H2 or CH3NHC+H2

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the major products obtained from the reaction of one equivalent of HCl with the following compounds. For each reaction, indicate the kinetic and thermodynamic products.
a. 2,3-dimethyl-1,3-pentadiene
b. 2,4-dimethyl-1,3-pentadiene
On a single graph, draw the reaction coordinate diagram for the addition of one equivalent of HBr to 2 methyl-1,3-pentadiene and for the addition of one equivalent of HBr to 2-methyl-1,4-pentadiene. Which reaction is faster?
Which dienophile in each pair is more reactive in a Diels–Alder reaction?
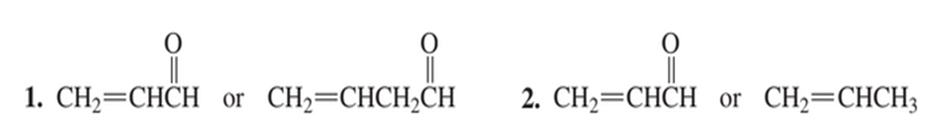
Which diene is more reactive in a Diels–Alder reaction?
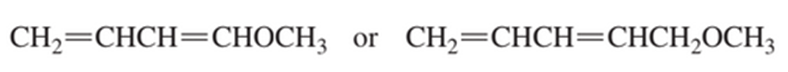
Which of the following is the strongest acid?
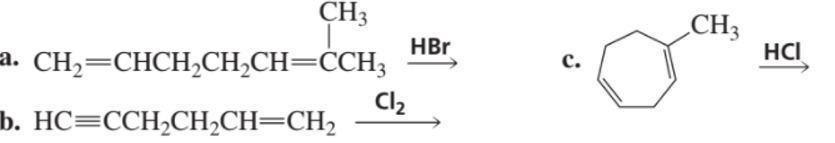
What is the major product of each of the following reactions, assuming that one equivalent of each reagent is used in each reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.