Chapter 1: Q52. (page 55)
Question: Draw all reasonable resonance structures for each species.
d. 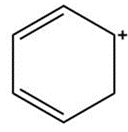
e.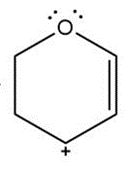
Short Answer
Answer
a.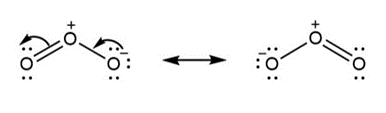
b.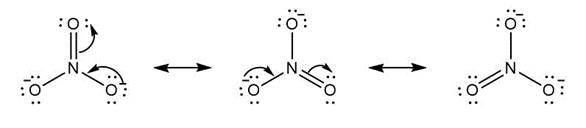
c.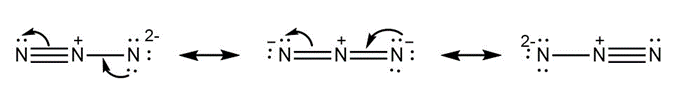
d.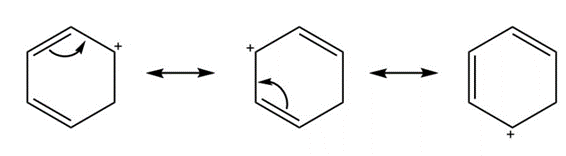
e.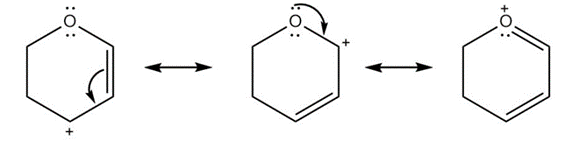
Learning Materials
Features
Discover
Chapter 1: Q52. (page 55)
Question: Draw all reasonable resonance structures for each species.
d. 
e.
Answer
a.
b.
c.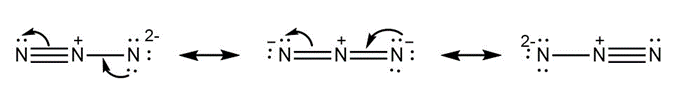
d.
e.
All the tools & learning materials you need for study success - in one app.
Get started for free
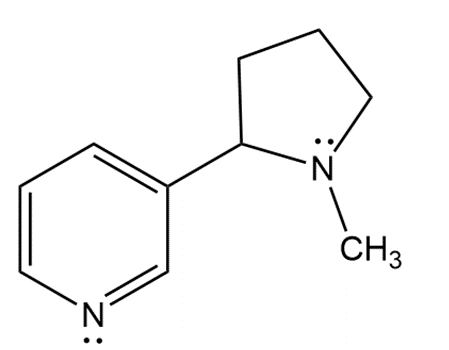
Question:
a. What is the hybridization of each N atom in nicotine?
b. What is the geometry around each N atom?
c. In what type of orbital does the lone pair on each N atom reside?
d. Draw a constitutional isomer of nicotine.
e. Draw a resonance structure of nicotine.
Question: Use the ball-and-stick model to answer each question about carbidopa, a drug used in combination with l-dopa to treat Parkinson’s disease.
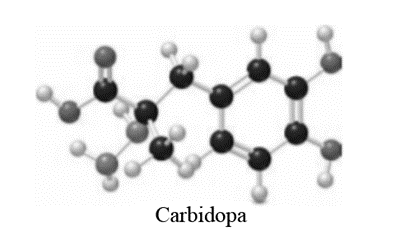
a. Draw a skeletal structure of carbidopa
b. Determine the hybridization around each carbon atom.
c. What is the hybridization and geometry around each N atom?
d. How many polar bonds are present?
Question: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.
a.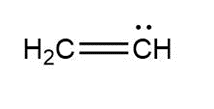
b. 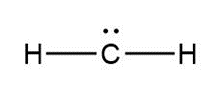
c.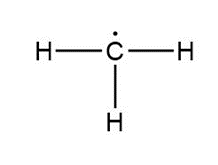
d.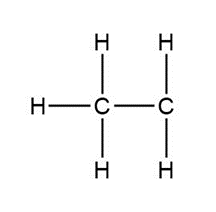
Question: Answer the following questions about acetonitrile :
a. Determine the hybridization of both C and N atoms.
b. Label all bonds as or .
c. In what type of orbital does the lone pair on N reside?
d. Label all bonds as polar or nonpolar.
Question: Label each bond in the following compounds as ionic or covalent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.