Chapter 5: Q.50 (page 208)
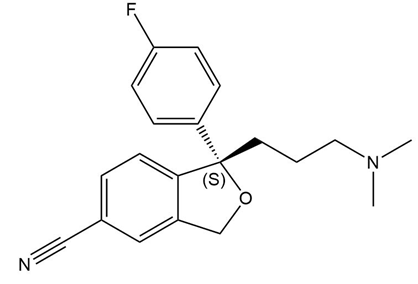
Draw the structure of (S)-citalopram, a drug used to treat depression and anxiety that is much more potent than its R enantiomer.
 citalopram
citalopram
Short Answer
The structure of the (S)-citalopram is as follows:
Learning Materials
Features
Discover
Chapter 5: Q.50 (page 208)
Draw the structure of (S)-citalopram, a drug used to treat depression and anxiety that is much more potent than its R enantiomer.
 citalopram
citalopram
The structure of the (S)-citalopram is as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw in a plane of symmetry for each molecule.
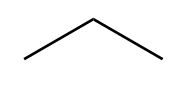
a.
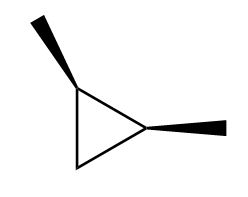
b.
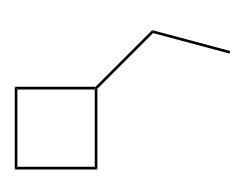
c.
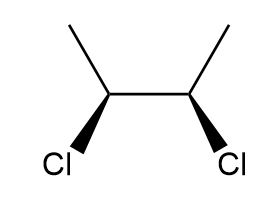
d.
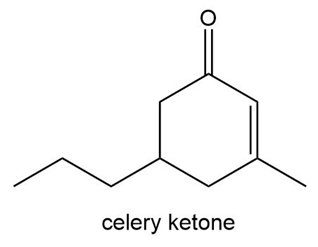
Like carvone, the two enantiomers of celery ketone smell differently. The R enantiomer smells like celery leaves, whereas the S enantiomer smells like licorice. Draw each enantiomer and assign its odor.
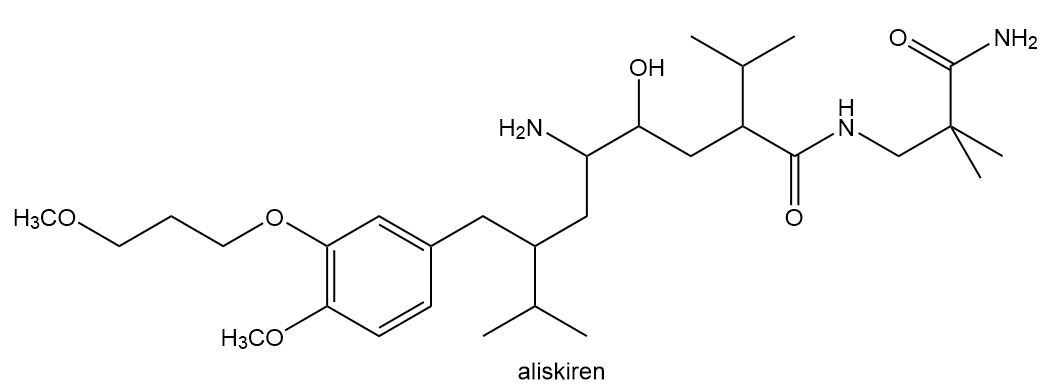
The facts in Section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in 2007 for the treatment of hypertension.
Explain each statement by referring to compounds A-E.
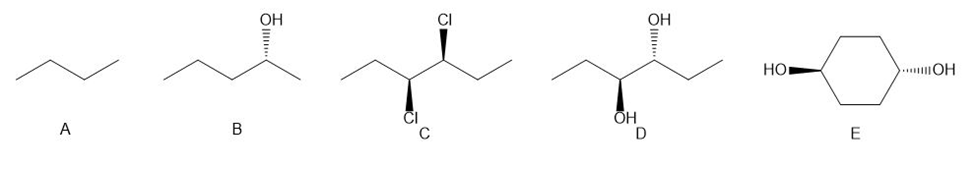
a. A has a mirror image but no enantiomer.
b. B has an enantiomer and no diastereomer.
c. C has both an enantiomer and a diastereomer.
d. D has a diastereomer but no enantiomer.
e. E has a diastereomer but no enantiomer.
a. Locate the stereogenic centres in the ball-and-stick model of lisinopril, a drug used to treat high blood pressure.
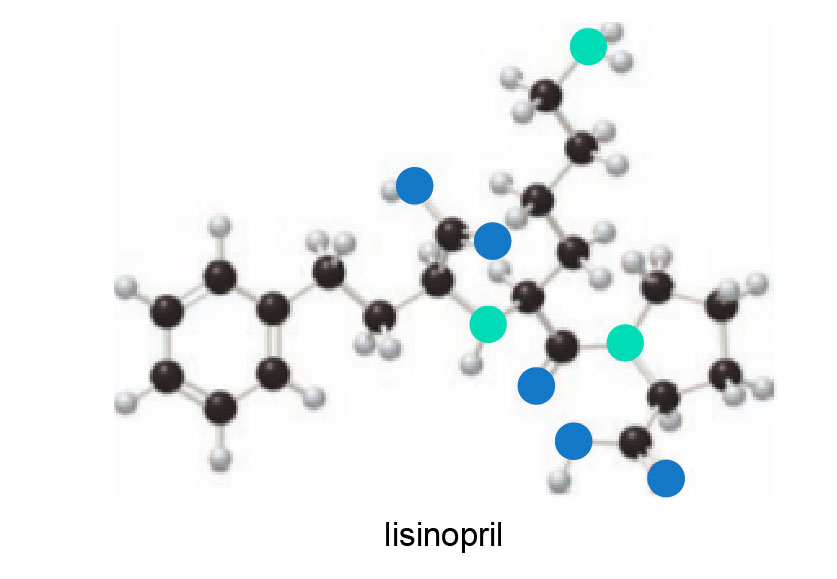
b. Label each stereogenic centre as R or S.
What do you think about this solution?
We value your feedback to improve our textbook solutions.