Chapter 5: Q.35 (page 204)
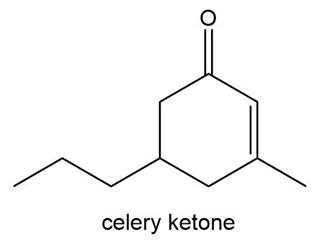
Like carvone, the two enantiomers of celery ketone smell differently. The R enantiomer smells like celery leaves, whereas the S enantiomer smells like licorice. Draw each enantiomer and assign its odor.
Short Answer
 Smells like celery leaves
Smells like celery leaves
 Smells like licorice
Smells like licorice
 Smells like celery leaves
Smells like celery leaves Smells like licorice
Smells like licorice