Chapter 5: Q.30 (page 200)
What is the ee for each of the following mixtures of enantiomers A and B?
a. 95% A and 5% B
b. 85% A and 15% B
Short Answer
a. The ee is 90%.
b. The ee is 70%.
Learning Materials
Features
Discover
Chapter 5: Q.30 (page 200)
What is the ee for each of the following mixtures of enantiomers A and B?
a. 95% A and 5% B
b. 85% A and 15% B
a. The ee is 90%.
b. The ee is 70%.
All the tools & learning materials you need for study success - in one app.
Get started for free
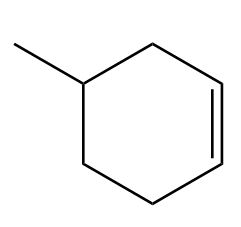
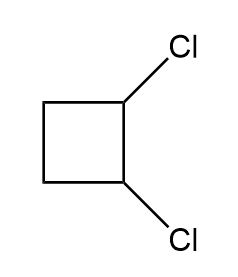
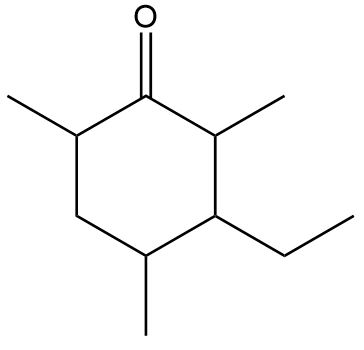
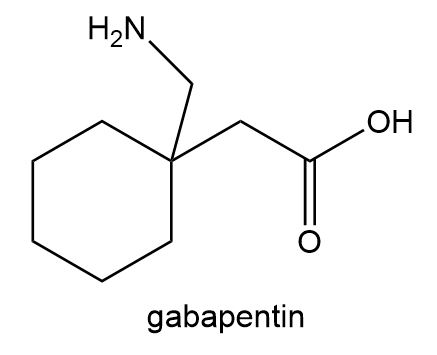
Locate the stereogenic centers in each compound. A molecule may have zero, one, or more stereogenic centers. Gabapentin [part (d)] is used clinically to treat seizures and certain types of chronic pain. Gabapentin enacarbil [part (e)] is a related compound that is three times more potent.
a.
b.
c.
d.
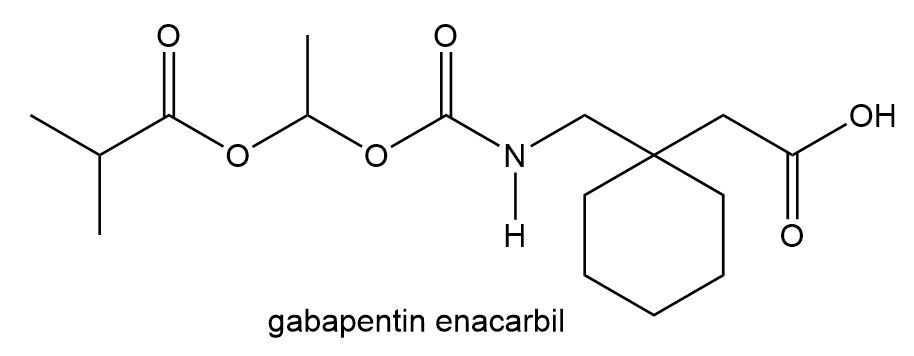
e.
f.
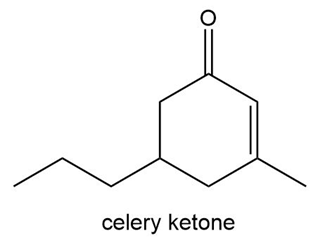
Like carvone, the two enantiomers of celery ketone smell differently. The R enantiomer smells like celery leaves, whereas the S enantiomer smells like licorice. Draw each enantiomer and assign its odor.
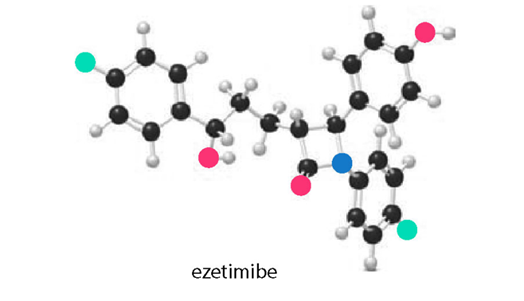
a) Locate the stereogenic centers in the ball-and-stick model of ezetimibe (trade name Zetia), a cholesterol-lowering drug. (b) Label each stereogenic center as R or S.
Locate the stereogenic centers in each compound.
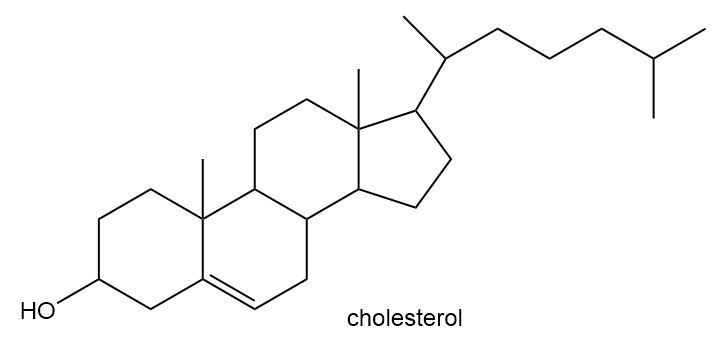
a.
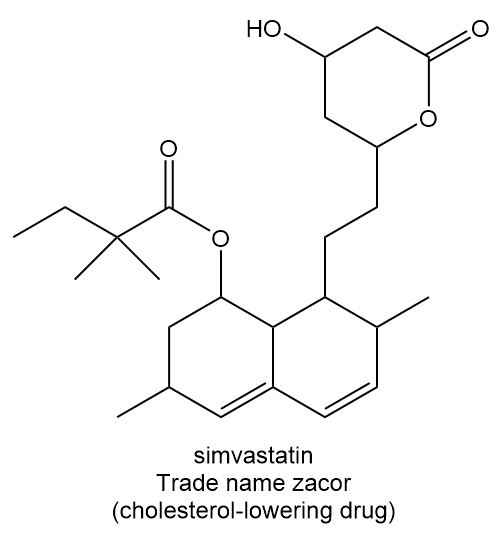
b.
Label the two stereogenic centers in each compound and draw all possible stereoisomers.
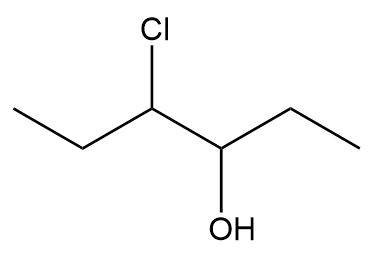
a.
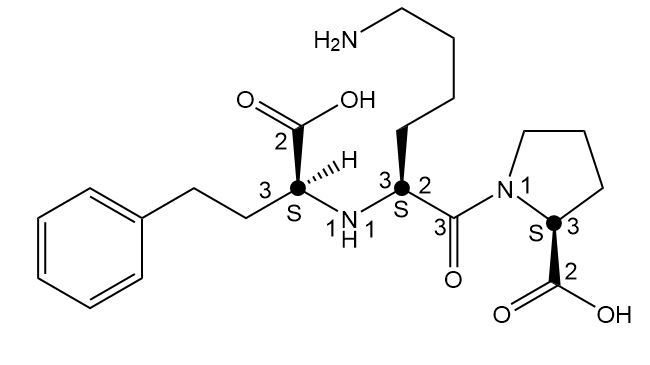
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.