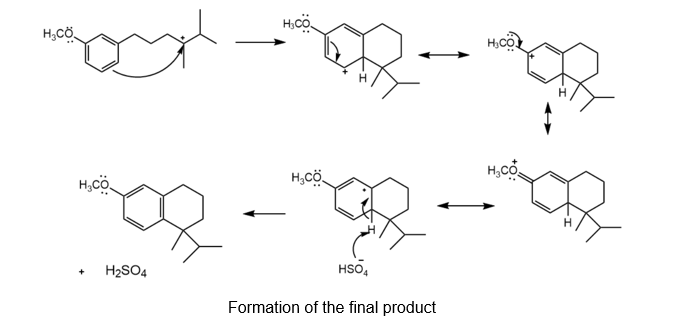
Chapter 18: Q 56. (page 724)
Question: Draw a stepwise, detailed mechanism for the following reaction.
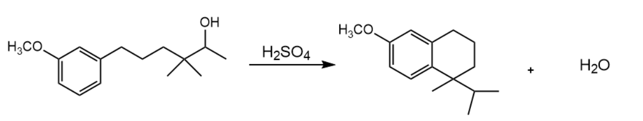
Short Answer
The detailed mechanism for the given reaction is as follows:
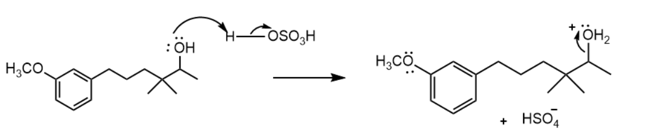
Protonation of the alcohol group
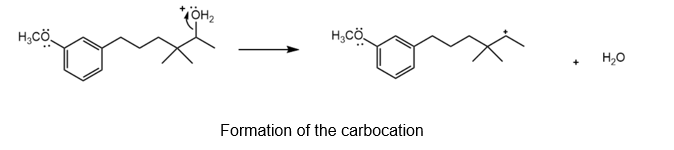
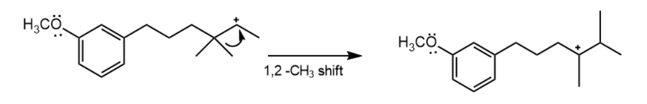
Formation of the tertiary carbocation
Learning Materials
Features
Discover
Chapter 18: Q 56. (page 724)
Question: Draw a stepwise, detailed mechanism for the following reaction.

The detailed mechanism for the given reaction is as follows:

Protonation of the alcohol group


Formation of the tertiary carbocation

All the tools & learning materials you need for study success - in one app.
Get started for free
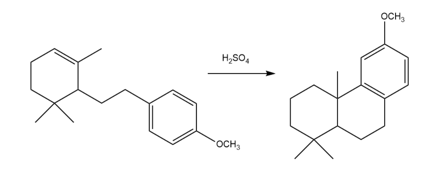
Question: Draw a stepwise, detailed mechanism for the following intramolecular reaction.
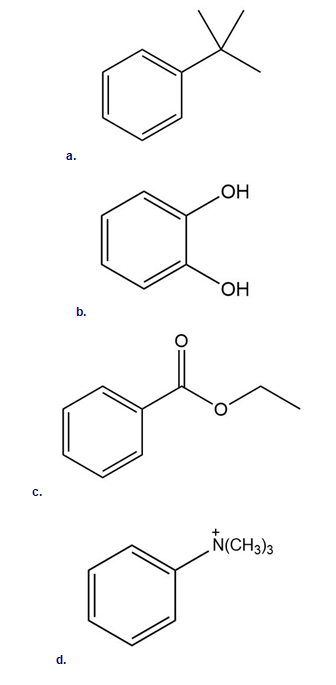
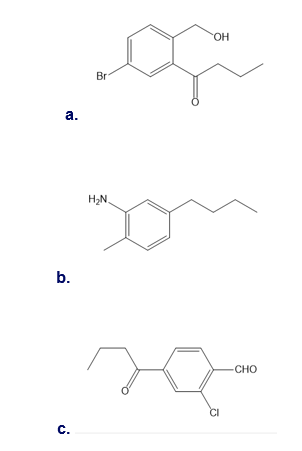
Label each compound as more or less reactive than benzene in electrophilic aromatic substitution.
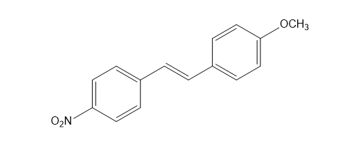
Question: What is the major product of electrophilic addition of HBr to the following alkene? Explain your choice.
Question: Rank the aryl halides in each group in order of increasing reactivity in nucleophilic aromatic substitution by an addition–elimination mechanism.
Question: Synthesize each compound from toluene and any other organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.