Chapter 18: 18-17P (page 677)
Label each compound as more or less reactive than benzene in electrophilic aromatic substitution.
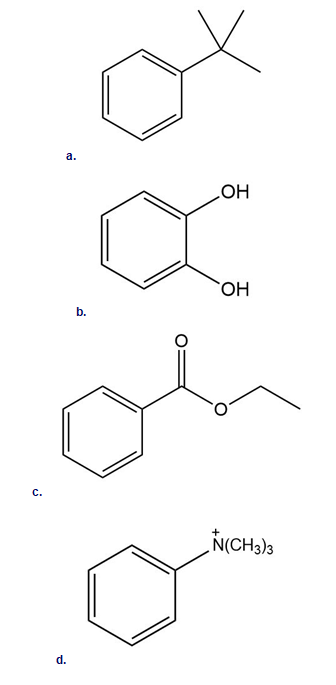
Short Answer
a. More reactive
b. More reactive
c. Less reactive
d. Less reactive
Learning Materials
Features
Discover
Chapter 18: 18-17P (page 677)
Label each compound as more or less reactive than benzene in electrophilic aromatic substitution.

a. More reactive
b. More reactive
c. Less reactive
d. Less reactive
All the tools & learning materials you need for study success - in one app.
Get started for free
Benzyl bromide\({\bf{(}}{{\bf{C}}_{\bf{6}}}{{\bf{H}}_{\bf{5}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{Br)}}\) reacts rapidly with \({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH}}\)to afford benzyl methyl ether\({\bf{(}}{{\bf{C}}_{\bf{6}}}{{\bf{H}}_{\bf{5}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{OC}}{{\bf{H}}_{\bf{3}}}{\bf{)}}\). Draw a stepwise mechanism for the reaction, and explain why this 1° alkyl halide reacts rapidly with a weak nucleophile under conditions that favor an\({{\bf{S}}_{\bf{N}}}{\bf{1}}\) mechanism. Would you expect the para-substituted benzylic halides \({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{O}}{{\bf{C}}_{\bf{6}}}{{\bf{H}}_{\bf{4}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{Br}}\) and \({{\bf{O}}_{\bf{2}}}{\bf{N}}{{\bf{C}}_{\bf{6}}}{{\bf{H}}_{\bf{4}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{Br}}\) to each be more or less reactive than benzyl bromide in this reaction?Explain your reasoning.
Question: Synthesize each compound from toluene and any other organic or inorganic reagents.
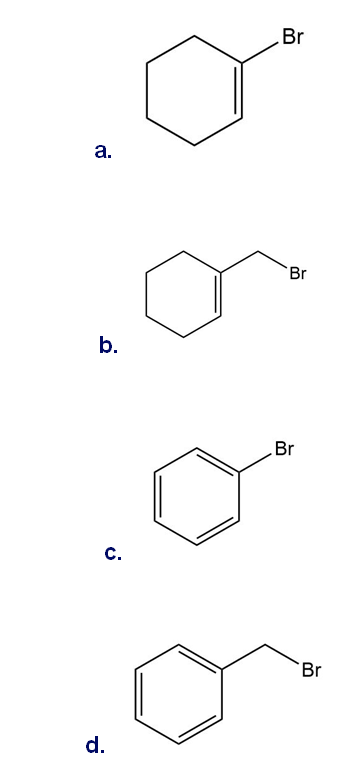
Which halides are unreactive in a Friedel–Crafts alkylation reaction?
Question: Draw a stepwise mechanism for the following reaction, which results in the synthesis of bisphenol F (R = H), an additive used in a variety of packaging materials. Bisphenol F is related to BPA (bisphenol A, R = ), a reagent used to harden some plastics, now removed from certain baby products because of its estrogen-like activity that can disrupt endocrine pathways.
What do you think about this solution?
We value your feedback to improve our textbook solutions.