Chapter 18: 18-7P (page 677)
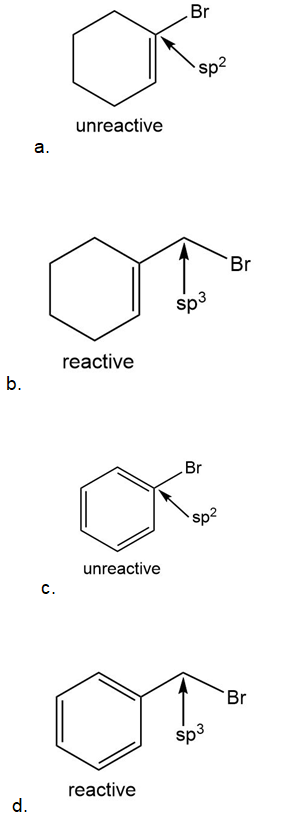
Which halides are unreactive in a Friedel–Crafts alkylation reaction?
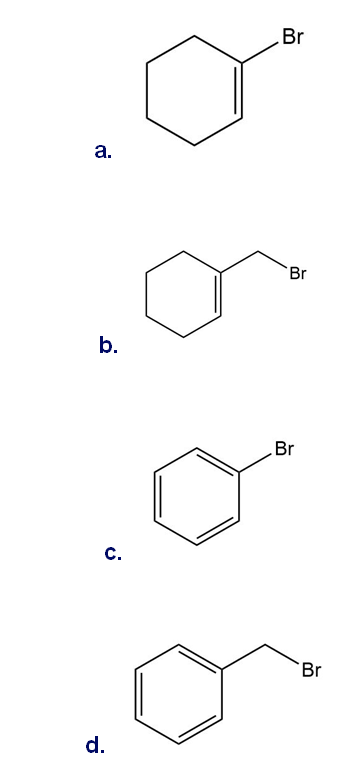
Short Answer
Expert verified
Learning Materials
Features
Discover
Chapter 18: 18-7P (page 677)
Which halides are unreactive in a Friedel–Crafts alkylation reaction?


All the tools & learning materials you need for study success - in one app.
Get started for free
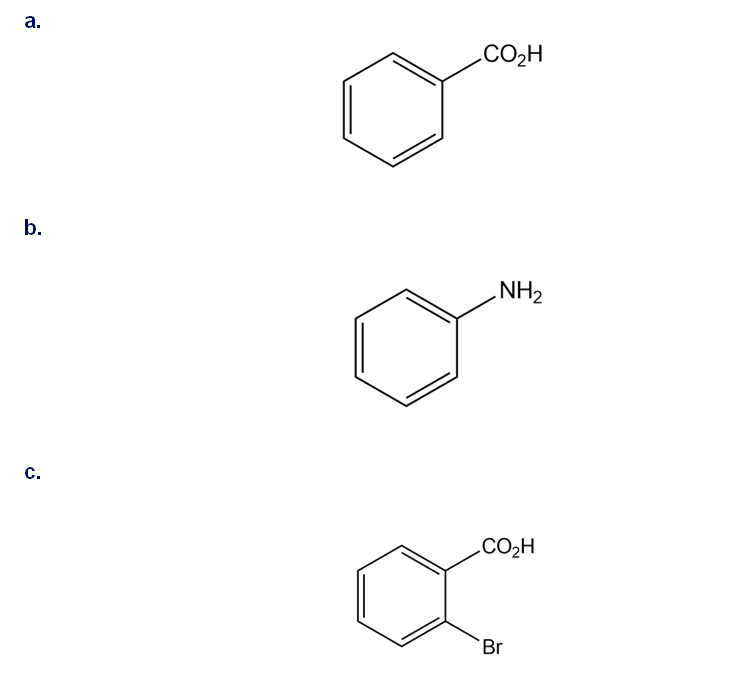
Question: Synthesize each compound from toluene and any other organic or inorganic reagents.
Question: Friedel-Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.
Question: Devise a synthesis of each compound from phenol and any other organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.