Chapter 18: Q 57. (page 724)
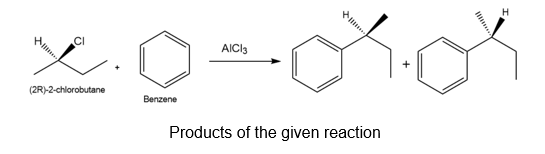
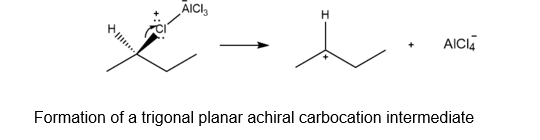
Question: Friedel-Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.
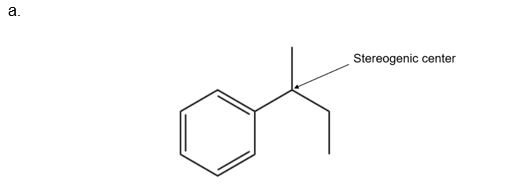
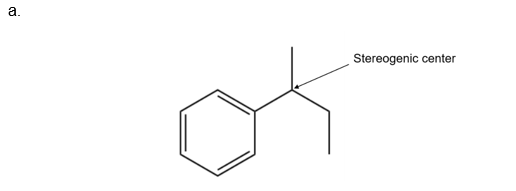
- How many stereogenic centers are present in the product?
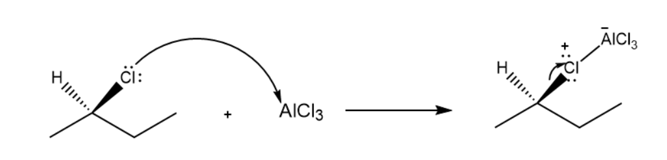
- Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.
Short Answer
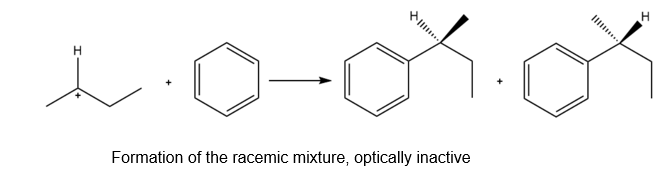
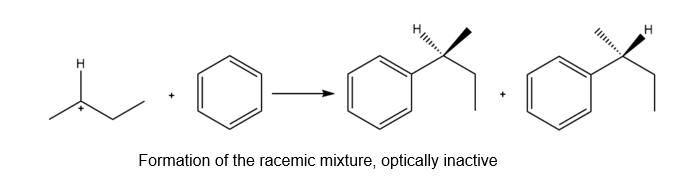
b. The reaction of the carbocation with the benzene results in the formation of a racemic mixture of two products which are optically inactive in nature as shown below: