Chapter 15: Q38. (page 570)
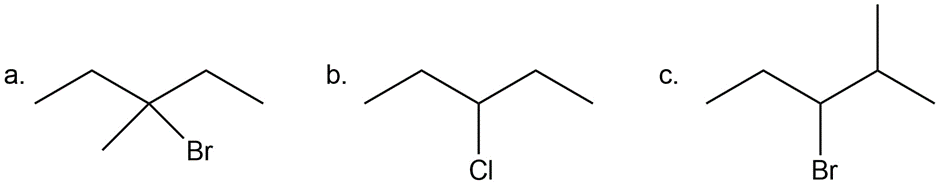
Question: Which alkyl halides can be prepared in good yield by radical halogenation of an alkane?
Short Answer
Answer
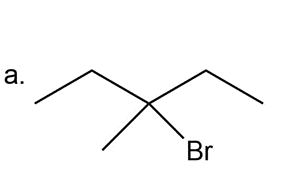
The tertiary alkyl halide (a) would be prepared in good yield by radical halogenation of an alkane.
Learning Materials
Features
Discover
Chapter 15: Q38. (page 570)
Question: Which alkyl halides can be prepared in good yield by radical halogenation of an alkane?

Answer
The tertiary alkyl halide (a) would be prepared in good yield by radical halogenation of an alkane.

All the tools & learning materials you need for study success - in one app.
Get started for free
Questions: Compounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are the structures of A and B?
Question: Draw the product(s) formed when each alkene is treated with either [1] HBr alone; or [2] HBr in the presence of peroxides.
a.
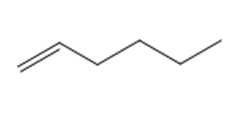
b.
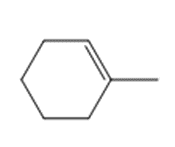
c.
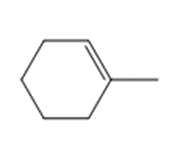
Question: Draw the organic products formed in each reaction.
a.
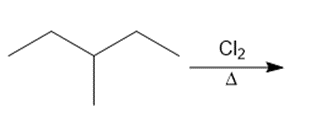
b.
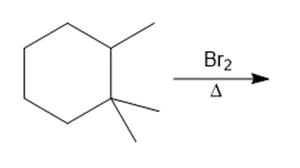
c.
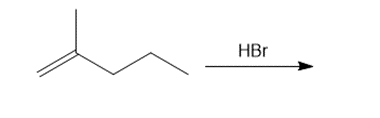
d.
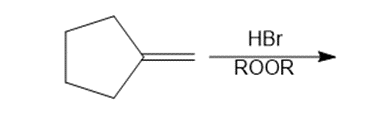
e.
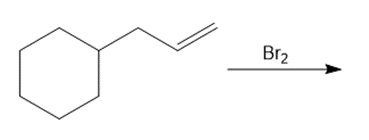
f.
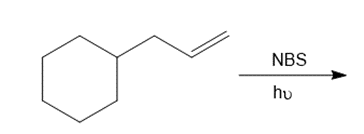
Question: As we will learn in Chapter 30, styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic polymerization is an example of electrophilic addition to an alkene involving carbocations.
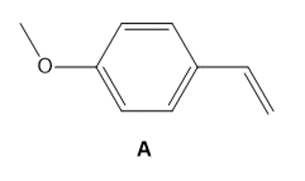
a. Draw a short segment of the polymer formed by the polymerization of A.
b. Why does A react faster than styrene (C6H5CH=CH2)in a cationic polymerization?
Question: Draw all resonance structures of the radical that results from abstraction of a hydrogen atom from the antioxidant BHA (butylated hydroxy anisole).
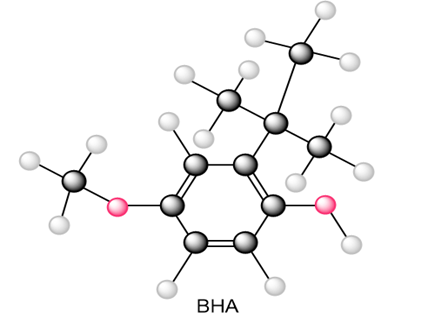
What do you think about this solution?
We value your feedback to improve our textbook solutions.