Chapter 14: Q.21558-14-46P (page 563)
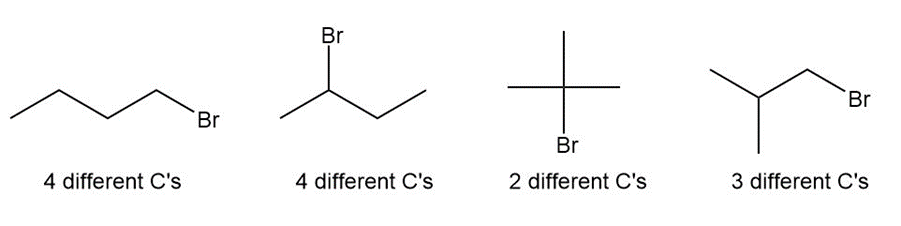
Question: Draw the four constitutional isomers having molecular formula C4H9Brand indicate how many different kinds of carbon atoms each has.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-46P (page 563)
Question: Draw the four constitutional isomers having molecular formula C4H9Brand indicate how many different kinds of carbon atoms each has.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
How many 1H NMR signals does each dimethylcyclopropane show?
Question: What effect does increasing the operating frequency of a1HNMR spectrum have on each value: (a) the chemical shift in ;(b) the frequency of an absorption in Hz; (c) the magnitude of a coupling constant J in Hz?
Question: The 1 H NMR spectrum of N,N-dimethylformamide shows three singlets at 2.9, 3.0, and 8.0 ppm. Explain why the two groups are not equivalent to each other, thus giving rise to two NMR signals.
For each compound give the number of NMR signals, and then determine how many peaks are present for each NMR signal.
a.
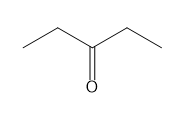
b.
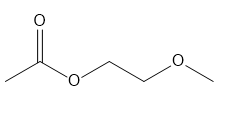
c.
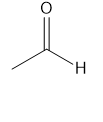
d.
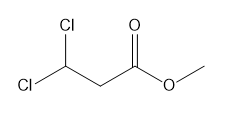
Rank each group of protons in order of increasing chemical shift.
a.
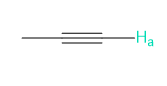
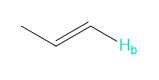
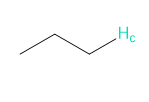
b.
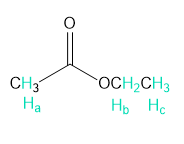
What do you think about this solution?
We value your feedback to improve our textbook solutions.