Chapter 14: Q17P (page 527)
Question: How many peaks are present in the NMR signal of each labelled proton?
a.
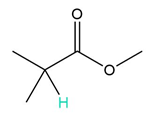
b.
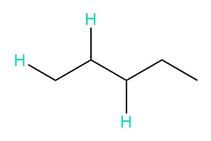
c.
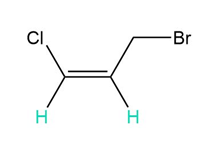
d.
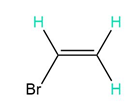
Short Answer
a. 7 peaks
b. 3, 5, 12, and 6 peaks
c. 2 and 6 peaks
d. 4, 4, and 4 peaks
Learning Materials
Features
Discover
Chapter 14: Q17P (page 527)
Question: How many peaks are present in the NMR signal of each labelled proton?
a.

b.

c.

d.

a. 7 peaks
b. 3, 5, 12, and 6 peaks
c. 2 and 6 peaks
d. 4, 4, and 4 peaks
All the tools & learning materials you need for study success - in one app.
Get started for free
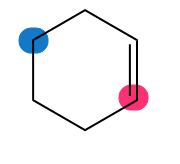
Which of the highlighted carbon atoms in each molecule absorbs farther downfield?
a.
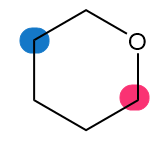
b.
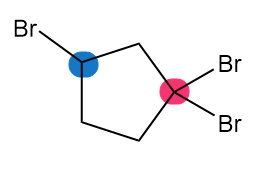
c.

d.
Question: Into how many peaks will the signal for each of the labeled protons be split?
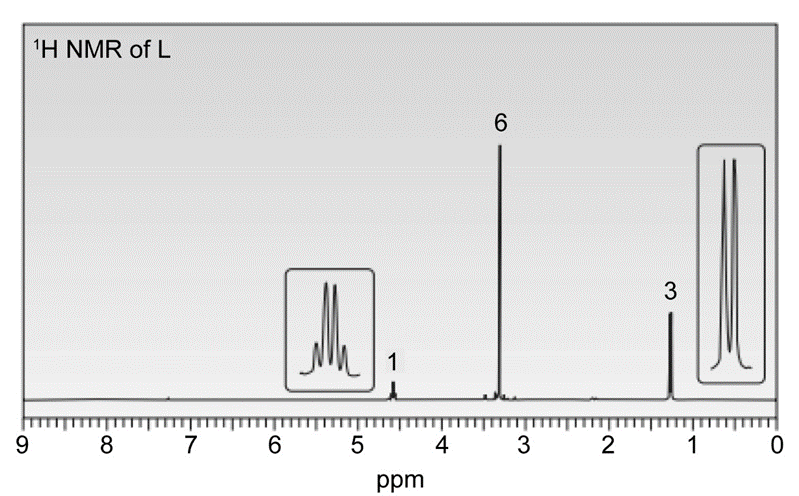
Question. In the presence of a small amount of acid, a solution of acetaldehyde(CH3CHO) in methanol (CH3OH) was allowed to stand and a new compound L was formed. L has a molecular ion in its mass spectrum at 90 and IR absorptions at 2992 and 2941cm-1 . L shows three signals in its 13C-NMR at 19, 52, and 101 ppm. 1H-NMR spectrum of L is given below. What is the structure of L?
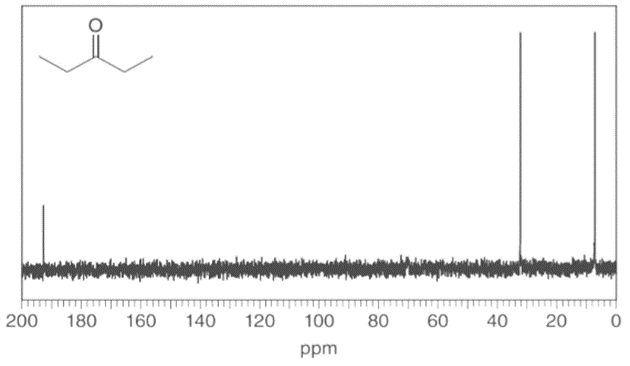
Question: Identify the carbon atoms that give rise to each NMR signal.
a.
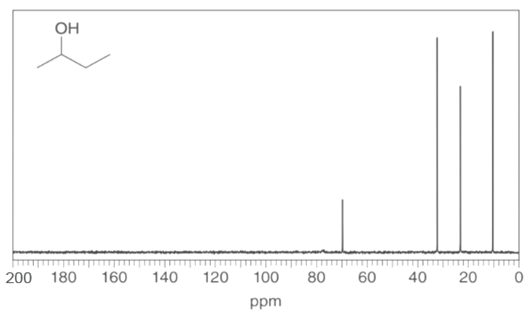
b
The 1H NMR spectrum 1,2-dimethoxyethane (CH3OCH2CH2OCH3) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?
What do you think about this solution?
We value your feedback to improve our textbook solutions.