Chapter 14: Q.21558-14-30P. (page 558)
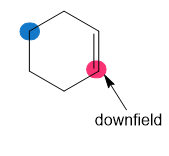
Which of the highlighted carbon atoms in each molecule absorbs farther downfield?
a.

b.
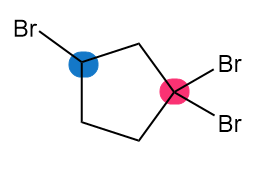
c.
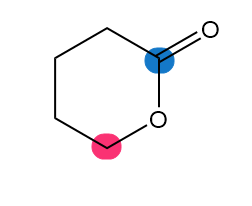
d.
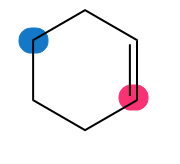
Short Answer
a.

b.

c.
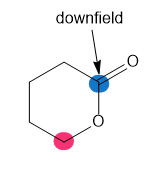
d.
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-30P. (page 558)
Which of the highlighted carbon atoms in each molecule absorbs farther downfield?
a.

b.

c.

d.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What effect does increasing the operating frequency of a1HNMR spectrum have on each value: (a) the chemical shift in ;(b) the frequency of an absorption in Hz; (c) the magnitude of a coupling constant J in Hz?
Question: Explain why the carbonyl carbon of an aldehyde or ketone absorbs farther downfield than the carbonyl carbon of an ester in a 13C NMR spectrum
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals does each dimethylcyclopropane show?
a. 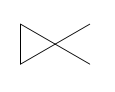
b. 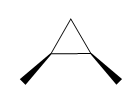
c.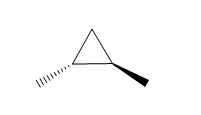
Question: Into how many peaks will the signal for each of the labeled protons be split?
What do you think about this solution?
We value your feedback to improve our textbook solutions.