Chapter 14: Q16P (page 527)
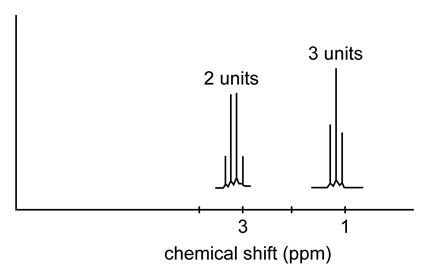
Question: Sketch the NMR spectrum of\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{Cl}}\), giving the approximate location of each NMR signal.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q16P (page 527)
Question: Sketch the NMR spectrum of\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{Cl}}\), giving the approximate location of each NMR signal.

All the tools & learning materials you need for study success - in one app.
Get started for free
The reaction of (CH3)3 CCHO with (C6H5)3 P=C(CH3)OCH3 , followed by treatment with aqueous acid, afford R . R has strong absorption in its IR spectrum at 1717 cm-1 and three singlets in its -NMR spectrum at 1.02 (9 H), 2.13 (3 H), and 2.33 (2 H) ppm. What is the structure of R? We will learn about this reaction in Chapter 21.
Question: Identify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound.
a. CH3CH2CH2CH2OH ; 13CNMR: 14, 19, 35, and 62 ppm
b. (CH3)2CHCHO ; 13C NMR: 16, 41, and 205 ppm
c. CH2=CHCHOHCH3 ; 13C NMR: 23, 69, 113, and 143 ppm
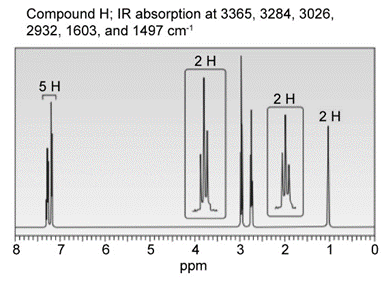
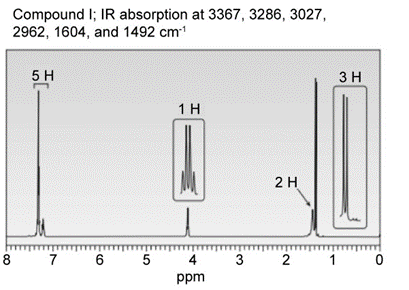
Question: Identify the structures of isomers H and I (molecular form C8H11N)
a.
b.
Question: Explain why the 13C NMR spectrum of 3-methylbutan-2-ol shows five signals.
How many NMR signals does each compound give?
a.
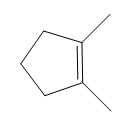
b.
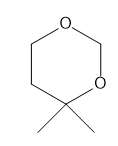
c.
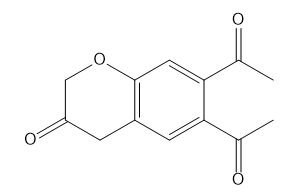
d.
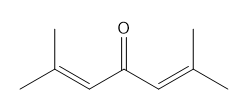
What do you think about this solution?
We value your feedback to improve our textbook solutions.