Chapter 3: Q.14. (page 91)
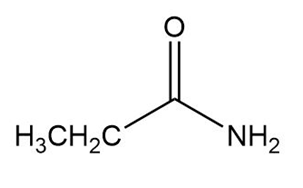
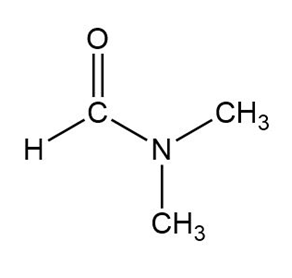
Question: Why the boiling point of propanamide , is considerably higher than the boiling point of N,N-dimethylformamide, ( vs. ), even though both compounds are isomeric amides.
Short Answer
Answer
The boiling point of propanamide is higher because of its ability to form hydrogen bonds. N,N-dimethylformamide is not able to form hydrogen bonds.