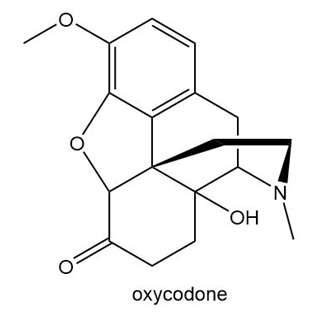
Chapter 3: Q.39. (page 91)
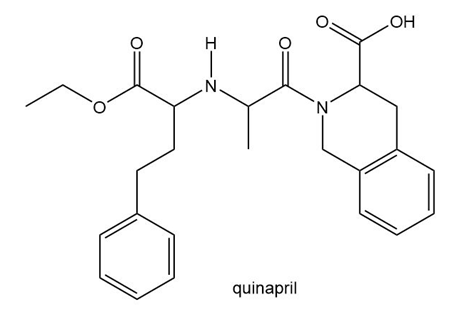
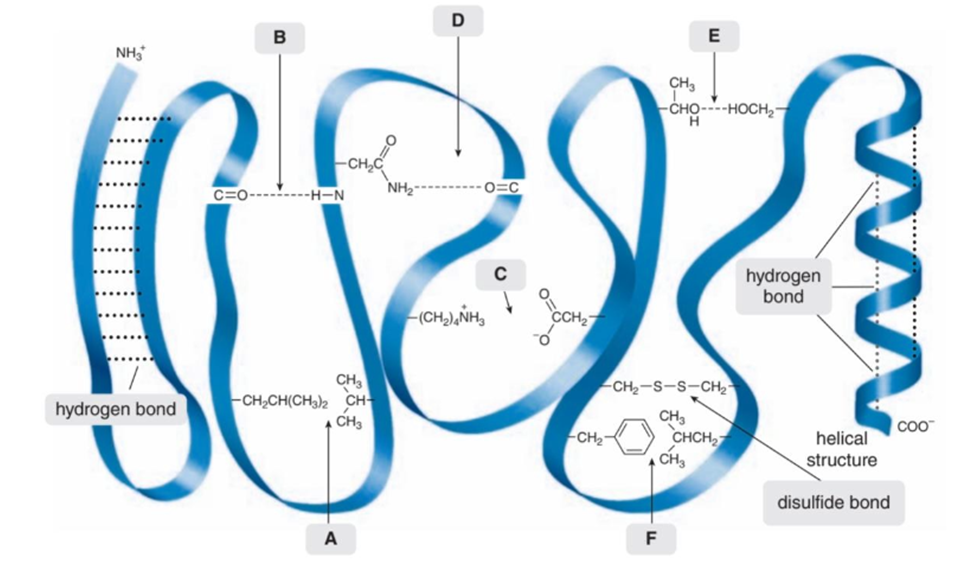
Question: Intramolecular forces of attraction are often important in holding large molecules together. For example, some proteins fold into compact shapes, held together by attractive forces between nearby functional groups. A schematic of a folded protein is drawn here, with the protein backbone indicated by a blue-green ribbon, and various appendages drawn dangling from the chain. What types of intramolecular forces occur at each labeled site (A–F)?
Short Answer
Answer
The intermolecular force present at site A is Vander Waals forces, site B is hydrogen bonding, site C is ion-ion interaction and Vander Waals forces, site D is hydrogen bonding, site E is Vander Waals forces, and site F is Vander Waals forces.