Chapter 3: Q.13. (page 91)
Question: Which compound in each pair has the higher boiling point?
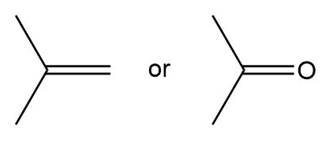
a.
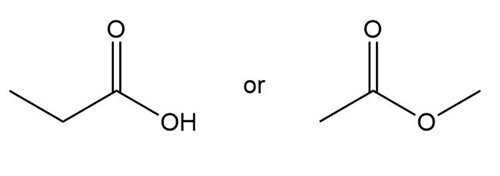
b.
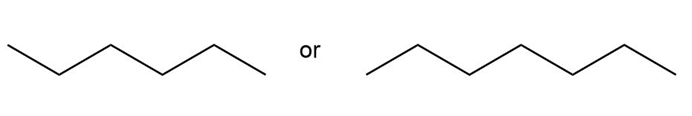
c.
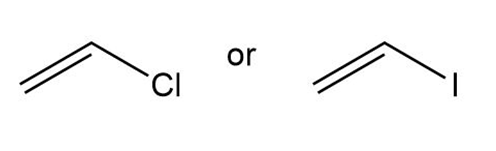
d.
Short Answer
Answer
The compounds that have higher boiling points are the following:
a.
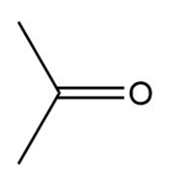
Propanone
b.
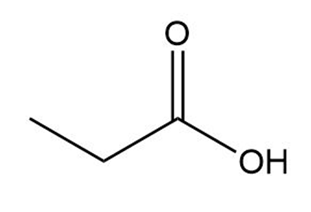
Propanoic acid
c.
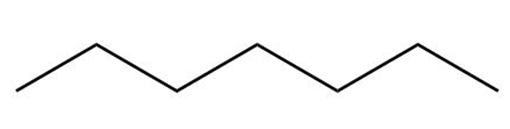
Heptane
d.
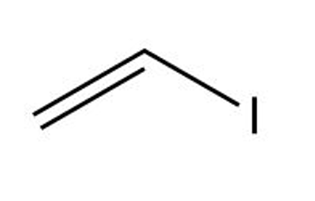
Vinyl iodide
Learning Materials
Features
Discover
Chapter 3: Q.13. (page 91)
Question: Which compound in each pair has the higher boiling point?
a.

b.

c.

d.

Answer
The compounds that have higher boiling points are the following:
a.

Propanone
b.

Propanoic acid
c.

Heptane
d.

Vinyl iodide
All the tools & learning materials you need for study success - in one app.
Get started for free
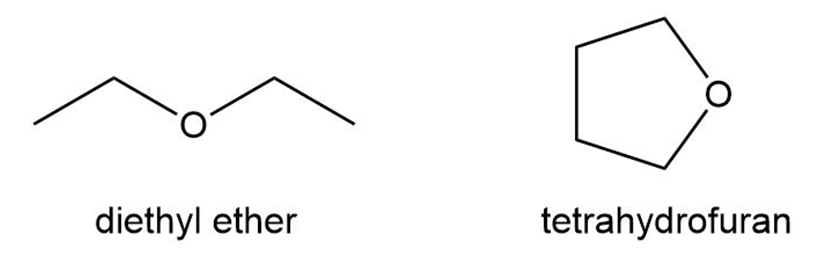
Question: Although diethyl ether and tetrahydrofuran are both four-carbon ethers, one compound is much more water soluble than the other. Predict which compound has higher water solubility and offer an explanation.
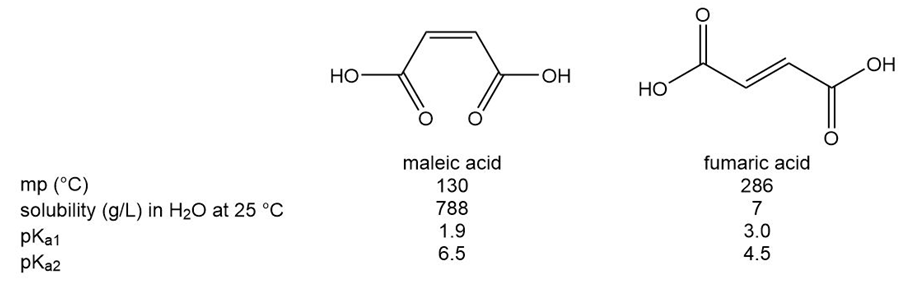
Question: Recall from Section 1.10B that there is restricted rotation around carbon-carbon double bonds. Maleic acid and fumaric acid are two isomers with vastly different physical properties and values for loss of both protons. Explain why each of these differences occurs.
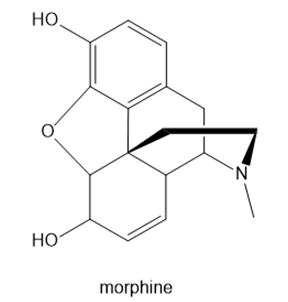
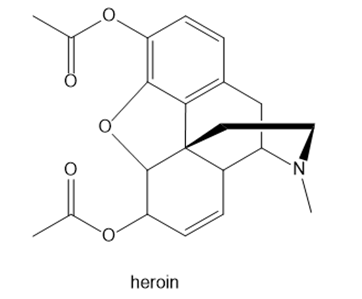
Question: (a) What types of intermolecular forces do morphine and heroin each possess? (b) Which compound can cross the blood–brain barrier more readily, and therefore serve as the more potent pain reliever?
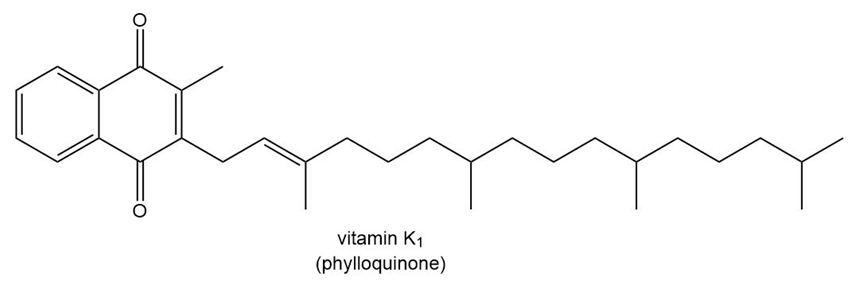
Question: Predict the water solubility of each vitamin.
a.
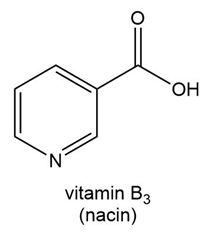
b.
Question: Identify each functional group located in the following rings. Which structure represents a lactone-a cyclic ester- and which represents a lactam-a cyclic amide?
a.
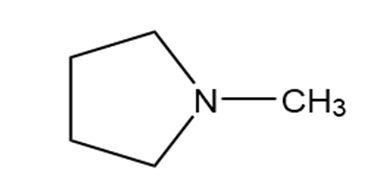
b.
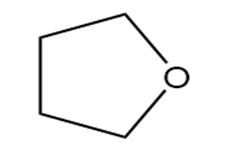
c.
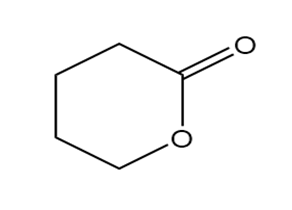
d.
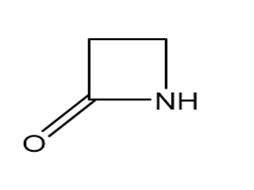
What do you think about this solution?
We value your feedback to improve our textbook solutions.