Chapter 3: Q.1. (page 91)
Question: What reaction occurs when is treated with a. ? b. NaH? What happens when is treated with these same reagents?
Short Answer
a.
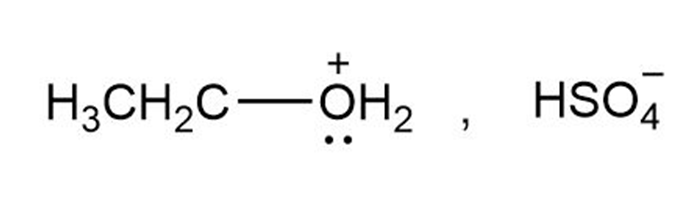
no reaction
b.
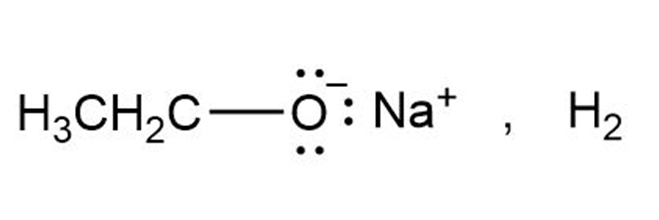
role="math" localid="1648118704086" no reaction
Learning Materials
Features
Discover
Chapter 3: Q.1. (page 91)
Question: What reaction occurs when is treated with a. ? b. NaH? What happens when is treated with these same reagents?
a.

no reaction
b.

role="math" localid="1648118704086" no reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
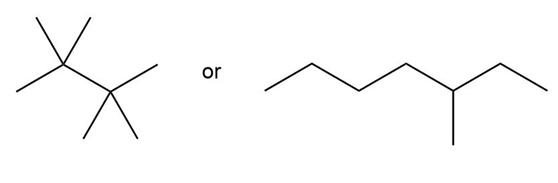
Question: Predict which compound in each pair has the higher melting point.
a.
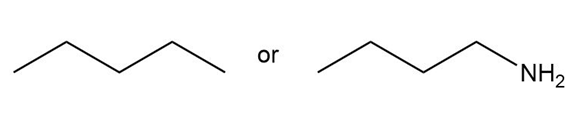
b.
Question: Draw the structure of a compound of molecular formula that fits each description: a. a compound that contains a amine and a alcohol; b. a compound that contains a amine and a alcohol.
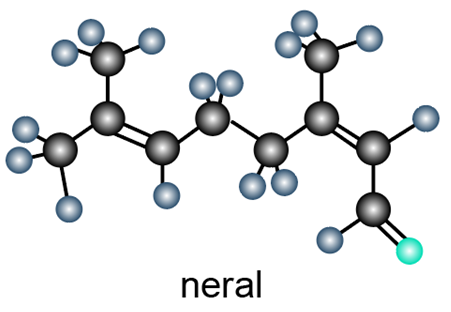
Question: a. Identify the functional groups in the ball-and-stick model of neral, a compound with a lemony odor isolated from lemon grass.
b. Draw a skeletal structure of a constitutional isomer of neral that should be more water soluble.
c. Label the most electrophilic carbon atom.
Question: Now that you have learned about solubility, explain why aspirin (Section 2.7) crosses a cell membrane as a neutral carboxylic acid rather than an ionic conjugate base.
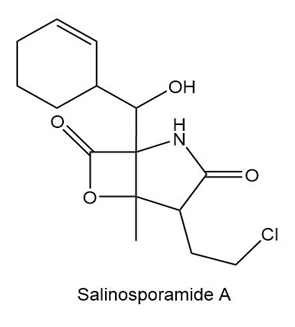
Question: (a)Identify the functional groups in salinosporamide A, an anticancer agent isolated from marine sediment.(b)Classify each alcohol, alkyl halide, amide, and amine as ,, or .
What do you think about this solution?
We value your feedback to improve our textbook solutions.