Chapter 3: Q.7. (page 91)
Question: Draw the structure of a compound of molecular formula that fits each description: a. a compound that contains a amine and a alcohol; b. a compound that contains a amine and a alcohol.
Short Answer
Answer
a.
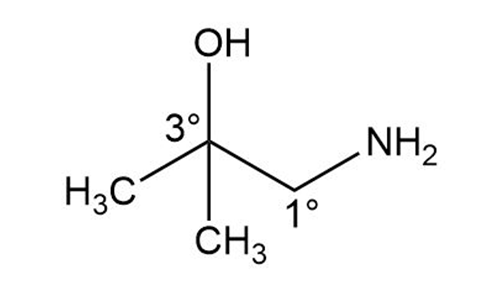
b.
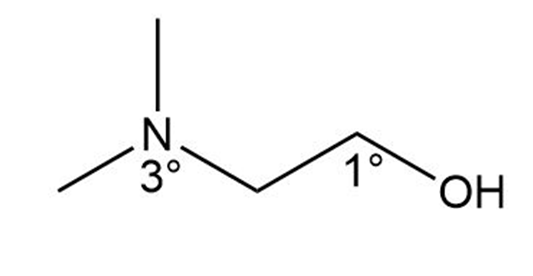
Learning Materials
Features
Discover
Chapter 3: Q.7. (page 91)
Question: Draw the structure of a compound of molecular formula that fits each description: a. a compound that contains a amine and a alcohol; b. a compound that contains a amine and a alcohol.
Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following molecules can hydrogen bond to another molecule of itself? Which can hydrogen bond with water?
a.
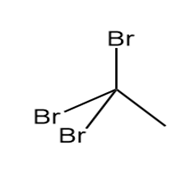
b.
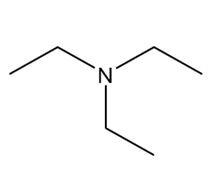
c.
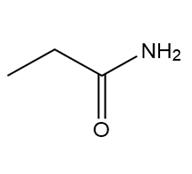
d.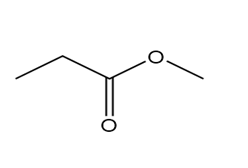
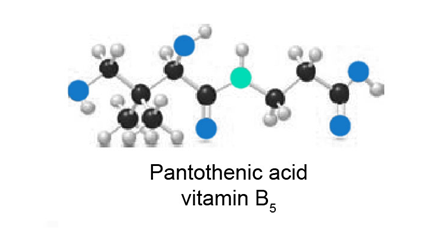
Question:(a) Identify the functional groups in the ball-and-stick model of pantothetic acid, vitamin .
(b) At which sites can pantothenic acid hydrogen bond to water?
(c) Predict the water solubility of pantothenic acid.
Question: Which compound in each pair has the higher boiling point?
a.
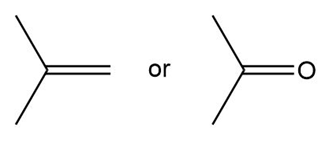
b.
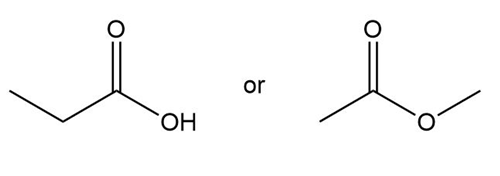
c.
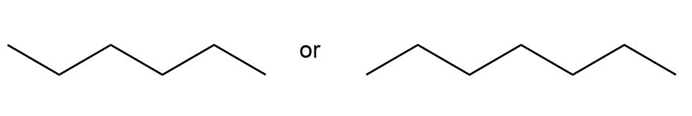
d.
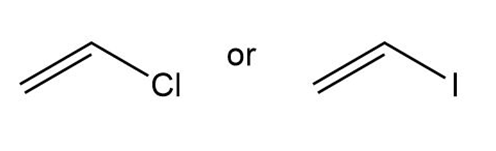
Question:(a)Draw four compounds with molecular formula , each containing at least one different functional group. (b) Predict which compound has the highest boiling point, and explain your reasoning.
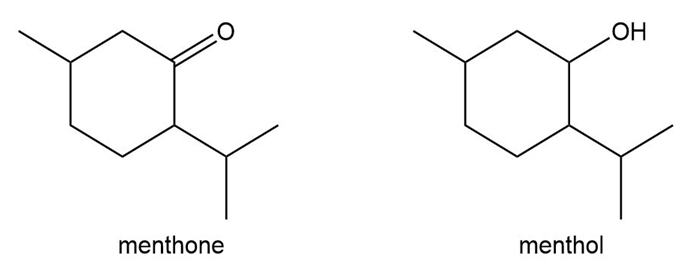
Question: Menthone and menthol are both isolated from mint. Explain why menthol is a solid at room temperature but menthone is a liquid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.