Chapter 16: PROBLEM 16.8 (page 612)
Question. Draw all possible resonance structures for the following cation and indicate which structure makes the largest contribution to the resonance hybrid.
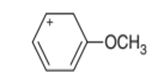
Short Answer
Answer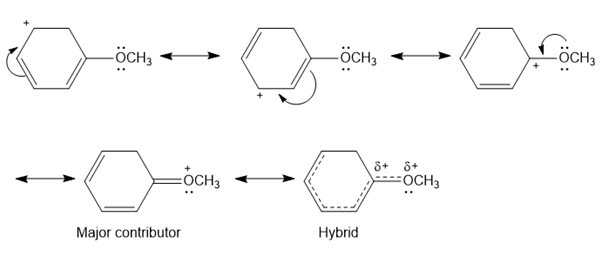
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.8 (page 612)
Question. Draw all possible resonance structures for the following cation and indicate which structure makes the largest contribution to the resonance hybrid.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
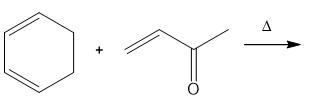
Question: Draw the products of the following Diels–Alder reactions. Indicate stereochemistry where appropriate.
a.
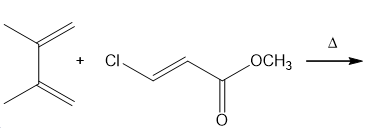
b.
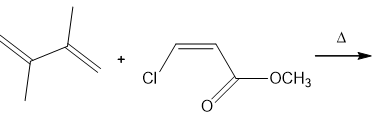
c.
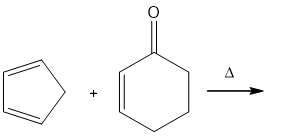
d.
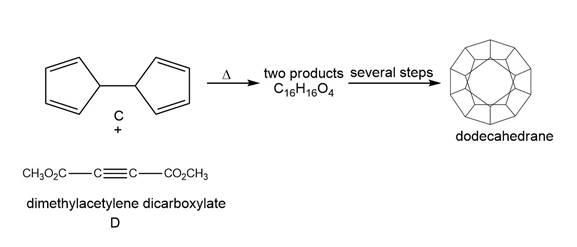
Question: One step in the synthesis of dodecahedrane (Section 4.11) involved reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4 . This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.
Question: Classify each diene as isolated or conjugated.
a.
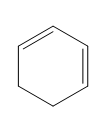
b.
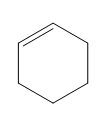
c.
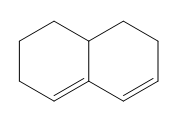
d.
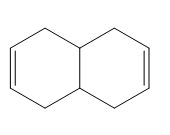
Question: Draw all possible stereoisomers of hepta-2,4-diene and label each double bond as E or Z.
Question: Devise a stepwise mechanism for the conversion of M to N. N has been converted in several steps to lysergic acid, a naturally occurring precursor of the hallucinogen LSD (Figure 18.4).
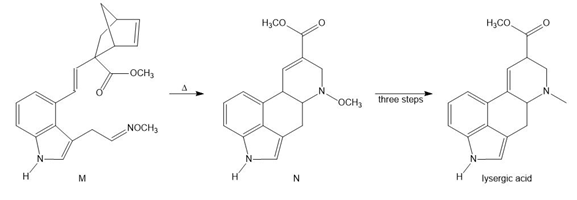
What do you think about this solution?
We value your feedback to improve our textbook solutions.