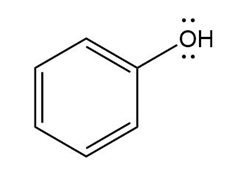
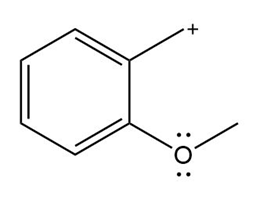
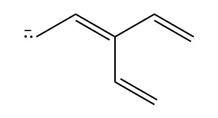
Chapter 16: PROBLEM 16.75 (page 640)
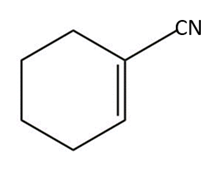
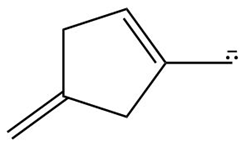
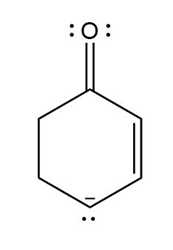
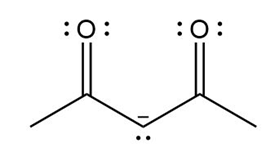
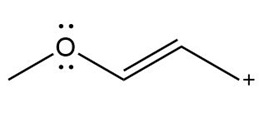
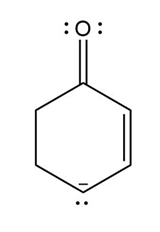
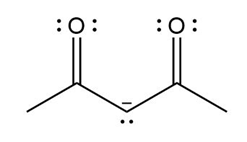
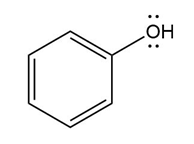
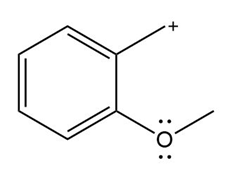
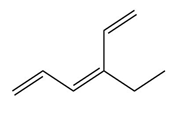
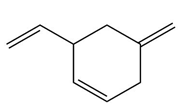
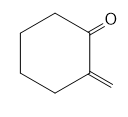
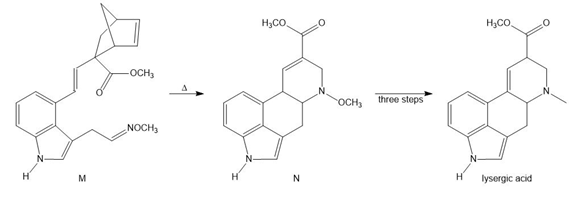
Question: Devise a stepwise mechanism for the conversion of M to N. N has been converted in several steps to lysergic acid, a naturally occurring precursor of the hallucinogen LSD (Figure 18.4).
Short Answer
Answer
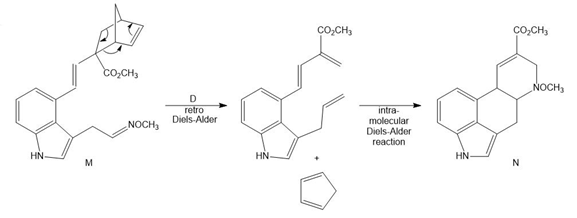
 Retro Diels-Alder product
Retro Diels-Alder product