Chapter 16: PROBLEM 16.66 (page 639)
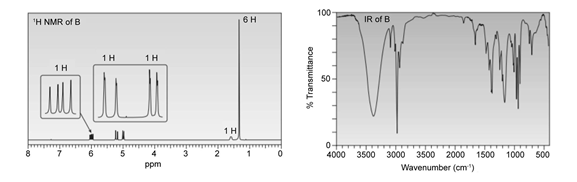
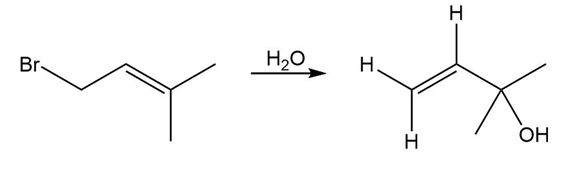
Question: The treatment of with forms B (molecular formula ) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.66 (page 639)
Question: The treatment of with forms B (molecular formula ) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Classify each diene as isolated or conjugated.
a.
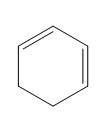
b.
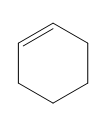
c.
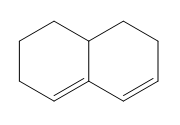
d.
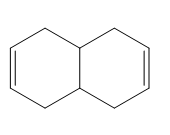
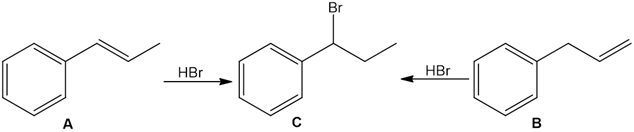
Question: Treatment of alkenes A and B with HBr gives the same alkyl halide C. Draw a mechanism for each reaction, including all reasonable resonance structures for any intermediate.
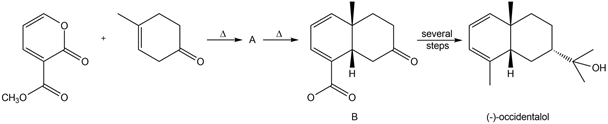
Question: One step in the synthesis of occidentalol, a natural product isolated from the eastern white cedar tree, involved the following reaction. Identify the structure of A and show how A is converted to B.
Question: Draw the products formed when each compound is treated with one equivalent of HBr.
a.
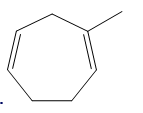
b.
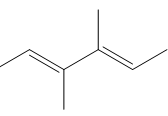
c.
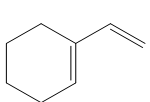
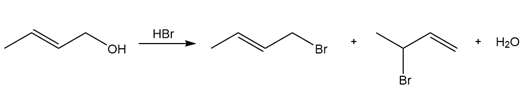
Question: Draw a stepwise mechanism for the following reaction
What do you think about this solution?
We value your feedback to improve our textbook solutions.