Chapter 16: PROBLEM 16.59 (page 638)
Question: Draw a stepwise mechanism for the following reaction
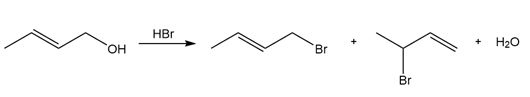
Short Answer
Answer
 Mechanism of reaction
Mechanism of reaction
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.59 (page 638)
Question: Draw a stepwise mechanism for the following reaction

Answer
 Mechanism of reaction
Mechanism of reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use resonance theory and the Hammond postulate to explain why 3-chloroprop-1-ene is more reactive than 1-chloropropane in SN1 reactions.
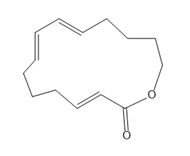
Question: A transannular Diels–Alder reaction is an intramolecular reaction that occurs when the diene and dienophile are contained in one ring, resulting in the formation of a tricyclic ring system. Draw the product formed when the following triene undergoes a transannular Diels–Alder reaction.
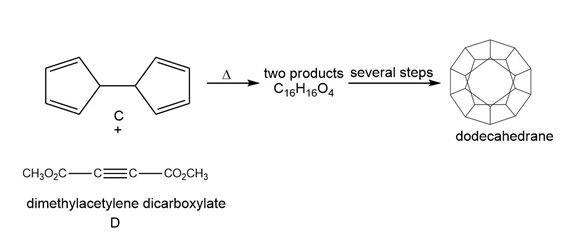
Question: One step in the synthesis of dodecahedrane (Section 4.11) involved reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4 . This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.
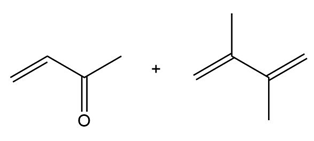
Question: Draw the product formed when each diene and dienophile react in a Diels–Alder reaction.
a.
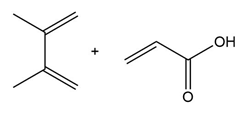
b.
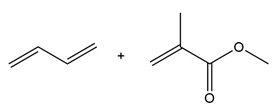
c.
Question: Label each pair of compounds as stereoisomers or conformations.
a.
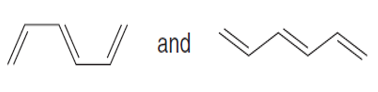
b.
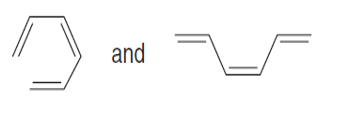
c.
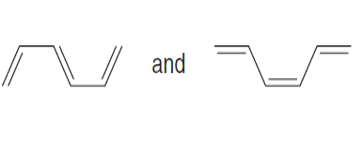
What do you think about this solution?
We value your feedback to improve our textbook solutions.