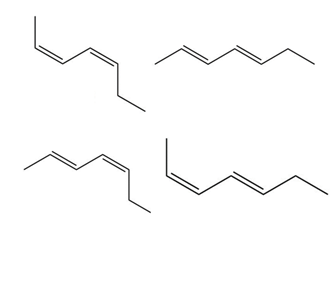
Chapter 16: PROBLEM 16.38 (page 635)
Question: Draw all possible stereoisomers of hepta-2,4-diene and label each double bond as E or Z.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.38 (page 635)
Question: Draw all possible stereoisomers of hepta-2,4-diene and label each double bond as E or Z.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
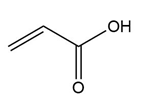
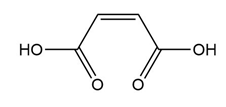
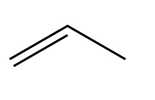
Question: Rank the following dienophiles in order of increasing reactivity
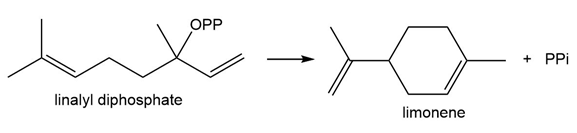
Question: Draw a stepwise mechanism for the biological conversion of linalyl diphosphate to limonene.
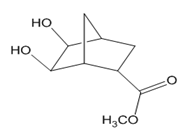
Question: Devise a stepwise synthesis of each compound from dicyclopentadiene using a Diels–Alder reaction as one step. You may also use organic compounds having ≤ 4 C’s, and any required organic or inorganic reagents:
a.
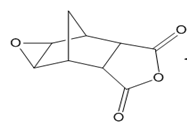
b.
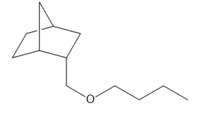
c.
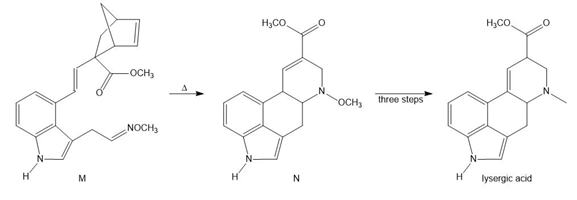
Question: Devise a stepwise mechanism for the conversion of M to N. N has been converted in several steps to lysergic acid, a naturally occurring precursor of the hallucinogen LSD (Figure 18.4).
Question: Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.