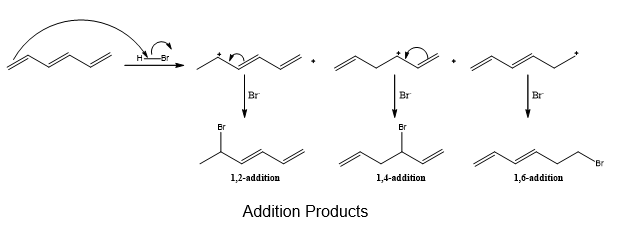
Chapter 16: PROBLEM 16.42 (page 635)
Question: Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.
Short Answer
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.42 (page 635)
Question: Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.

All the tools & learning materials you need for study success - in one app.
Get started for free
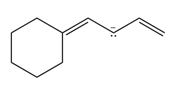
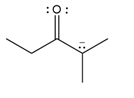
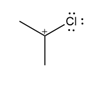
Question: Draw additional resonance structures for each ion.
a.
b.
c.
d.
Question: a. Draw resonance structures for the carbocation formed after loss of a leaving group from phytyl diphosphate. b. Draw the two-step mechanism for Friedel–Crafts alkylation of 1,2-dihydroxynaphthoic acid with this carbocation to form X.
Question: Rank the following compounds in order of increasing stability.
a.
b.
c.
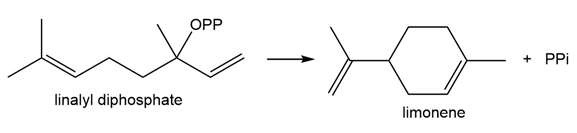
Question: Draw a stepwise mechanism for the biological conversion of linalyl diphosphate to limonene.
Question: a. Draw the two isomeric dienes formed when is treated with an alkoxide base.
b. Explain why the major product formed in this reaction does not contain the more highly substituted alkene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.