Chapter 16: PROBLEM 16.23 (page 625)
Question: Draw the products of each Diels–Alder reaction, and indicate the stereochemistry.
a.
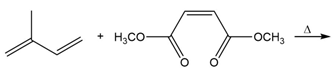
b.
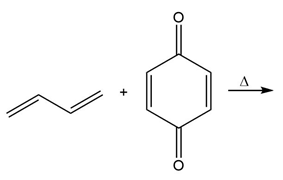
c.
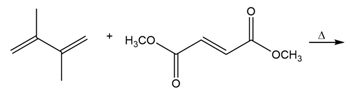
Short Answer
Answer
a.
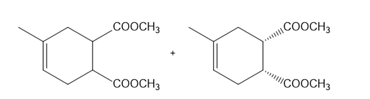
b.
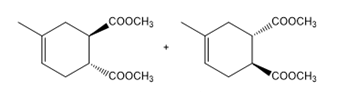
c.
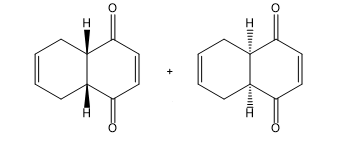
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.23 (page 625)
Question: Draw the products of each Diels–Alder reaction, and indicate the stereochemistry.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the products formed when each compound is treated with one equivalent of HBr.
a.
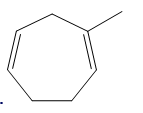
b.
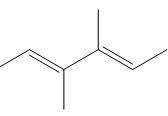
c.
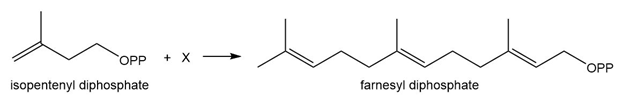
Question: Farnesyl diphosphate is synthesized from isopentenyl diphosphate and X by a pathway similar toMechanism 16.1. Draw the structure of X.
Question: Why is the bond dissociation energy for the C-C bond in ethane much higher than the bond dissociation energy for the labeled C-C in but-1-ene?
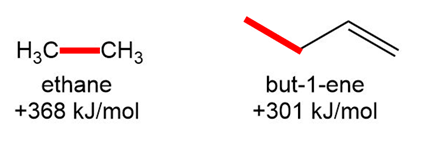
Question: Intramolecular Diels–Alder reactions are possible when a substrate contains both a 1,3-diene and a dienophile, as shown in the following general reaction.
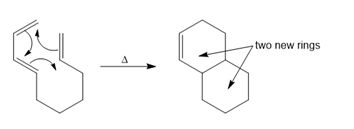
With this in mind, draw the product of each intramolecular Diels–Alder reaction.
a.
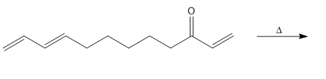
b.
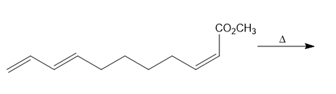
Question: Devise a stepwise synthesis of each compound from dicyclopentadiene using a Diels–Alder reaction as one step. You may also use organic compounds having ≤ 4 C’s, and any required organic or inorganic reagents:
a.
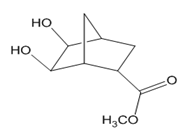
b.
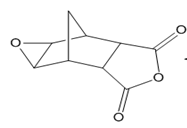
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.