Chapter 16: PROBLEM 16.5 (page 608)
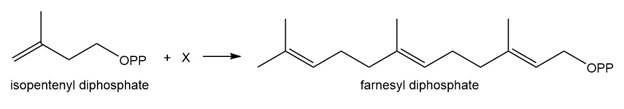
Question: Farnesyl diphosphate is synthesized from isopentenyl diphosphate and X by a pathway similar toMechanism 16.1. Draw the structure of X.
Short Answer
ANSWER
Learning Materials
Features
Discover
Chapter 16: PROBLEM 16.5 (page 608)
Question: Farnesyl diphosphate is synthesized from isopentenyl diphosphate and X by a pathway similar toMechanism 16.1. Draw the structure of X.

ANSWER

All the tools & learning materials you need for study success - in one app.
Get started for free
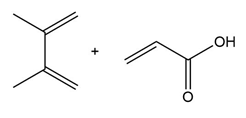
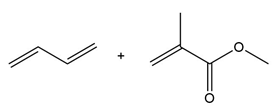
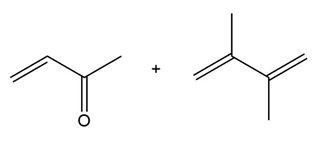
Question: Draw the product formed when each diene and dienophile react in a Diels–Alder reaction.
a.
b.
c.
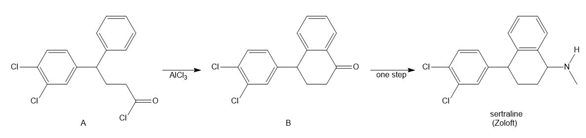
Question: a. Draw resonance structures for the carbocation formed after loss of a leaving group from phytyl diphosphate. b. Draw the two-step mechanism for Friedel–Crafts alkylation of 1,2-dihydroxynaphthoic acid with this carbocation to form X.
Question: Rank the following dienophiles in order of increasing reactivity
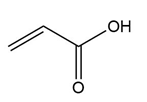
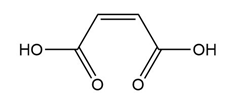
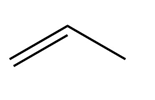
Question: Treatment of alkenes A and B with HBr gives the same alkyl halide C. Draw a mechanism for each reaction, including all reasonable resonance structures for any intermediate.
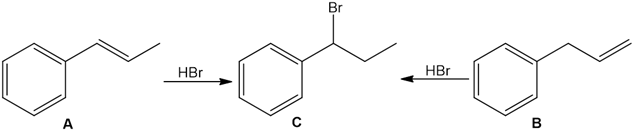
Question: A transannular Diels–Alder reaction is an intramolecular reaction that occurs when the diene and dienophile are contained in one ring, resulting in the formation of a tricyclic ring system. Draw the product formed when the following triene undergoes a transannular Diels–Alder reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.